New Publication from Ibáñez Lab: Sustained Anti-obesity Effects of Life-style Change and Anti-inflammatory Interventions after Conditional Inactivation of the Activin Receptor ALK7
Life-style change and anti-inflammatory interventions have only transient effects in obesity. It is not clear how benefits obtained by these treatments can be maintained in longer term, especially during sustained high caloric intake. Recently, a research from Carlos F. Ibáñez lab (“Sustained anti-obesity effects of life-style change and anti-inflammatory interventions after conditional inactivation of the activin receptor ALK7”) published on The FASEB Journal reveals that the constitutive ablation of the activin receptor ALK7 in adipose tissue enhances catecholamine signaling and lipolysis in adipocytes, and protects mice from diet-induced obesity. Although ALK7 deletion had little effect on its own, it synergized strongly with a transient switch to low-fat diet (life-style change) or anti-inflammatory treatment (Na-salicylate), resulting in enhanced lipolysis, increased energy expenditure, and reduced adipose tissue mass and body weight gain, even under sustained high caloric intake. By themselves, diet-switch and salicylate had only a temporary effect on weight gain. Mechanistically, combination of ALK7 ablation with either treatment strongly enhanced the levels of β3-AR, the main adrenergic receptor for catecholamine stimulation of lipolysis, and C/EBPα, an upstream regulator of β3-AR expression. These results suggest that inhibition of ALK7 can be combined with simple interventions (like diet adjustment or anti-inflammatory interventions) to produce longer-lasting benefits in obesity.
In this study, Ibáñez lab used a genetic approach to conditionally knockout ALK7 in adult obese mice, following 12 weeks on high-fat diet (HFD). They combined conditional ALK7 knockout with either life-style change (i.e., switch from high- to low-fat diet) or anti-inflammatory treatment (Na-salicylate), and assessed effects on weight gain, fat deposition and different biochemical and physiological parameters in several adipose tissue depots.
The study suggested that acute ablation of ALK7 in adipose tissue of obese mice enhances the beneficial effects of the switch to a low caloric diet. To assess a possible interaction between inactivation of ALK7 and life-style change interventions, a parallel cohort of HFD-fed control and mutant mice was switched to standard Chow diet (SD) 2 days after the last tamoxifen administration. Both control and mutant mice showed a steady drop in body weight after the diet switch, but weight loss occurred more rapidly and to a greater extent in the mutant mice. The expression level assessments of a battery of markers of adipose tissue differentiation, function and inflammation also indicates that ALK7 inactivation and diet switch synergize to restore catecholamine sensitivity in adipose tissue after nutrient overload. In addition, the examination of mRNA expression for a battery of inflammatory markers also revealed a marked reduction of inflammation in adipose tissues after diet switch.
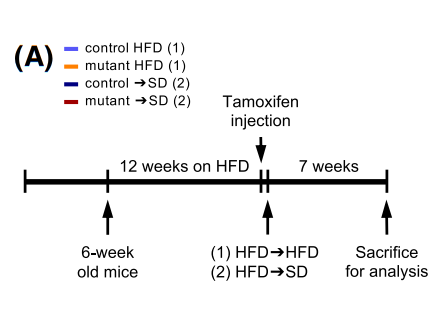
Figure 1 Schematic of the diet switch protocol.
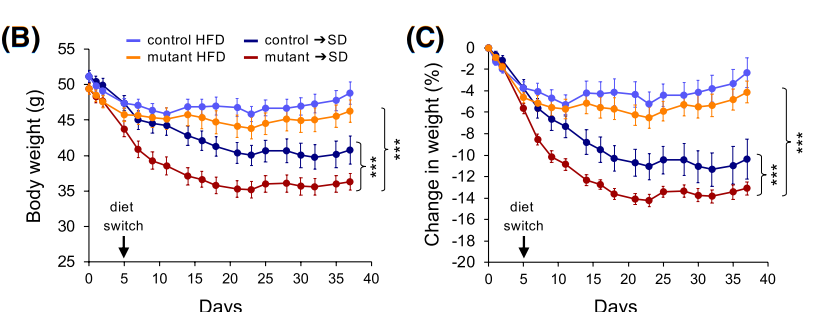
Figure 2 Body weights of control and mutant mice in constant high-fat diet (HFD) or after a switch to standard diet (→SD) as indicated.
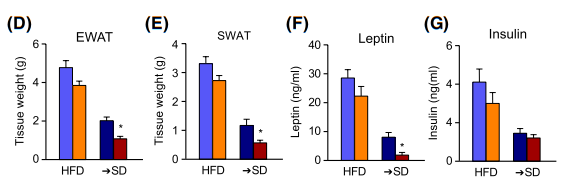
Figure 3 Weights of epididymal (EWAT) and subcutaneous (SWAT) white adipose tissues, and Plasma levels of leptin (EF) and insulin (G) in control and mutant mice after constant HFD or diet switch (→SD).
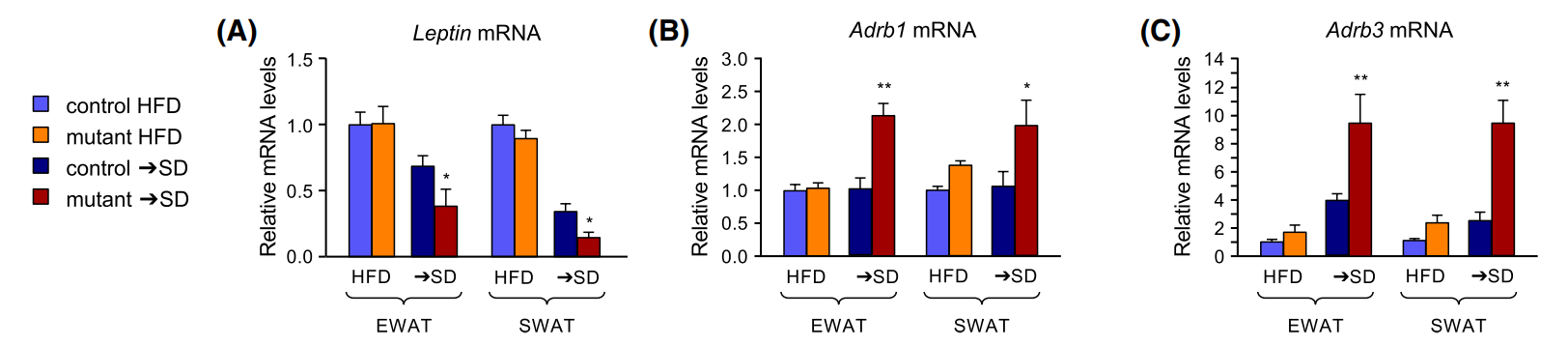
Figure 4 Q-PCR determination of Leptin (A), Adrb1 (B) and Adrb3 (C) mRNA expression in EWAT and SWAT of control and mutant mice after constant HFD or diet switch (→SD).
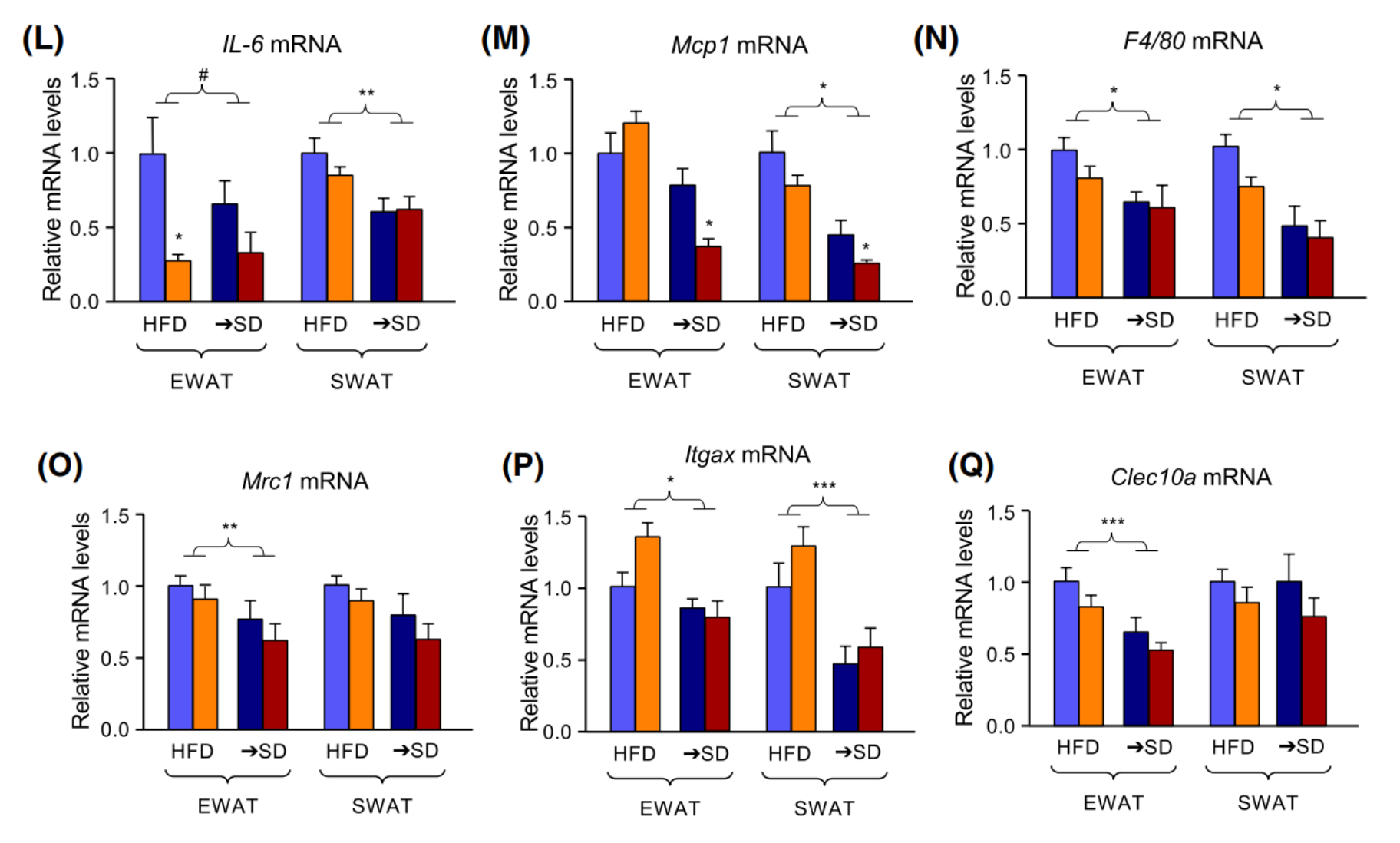
Figure 5 Determination of inflammation markers mRNA expression in EWAT and SWAT of control and mutant mice after constant HFD or diet switch (→SD).
The study showed that ALK7 ablation in adipose tissue of obese mice sustains the anti-obesity effects of diet switch after return to high caloric diet. In order to assess whether the beneficial effects of combined ALK7 ablation and diet switch had a longer lasting effect on metabolism, the lab subjected a separate cohort of mice to a double-switch paradigm, in which obese mice that had been switched to SD went back to HFD for additional 5 weeks. After these mice were switched back to HFD, control mice gained significantly more weight than mutant mice. On the other hand, mutant mice regained weight at a much slower pace, and were able to maintain a significant difference compared to the groups that remained under constant HFD. Assessment of other relevant markers (e.g. glucose tolerance and insulin sensitivity) also suggested that by opposing diet-induced catecholamine resistance, ALK7 ablation helped to sustain the anti-obesity effects of a diet switch, even after returning to a high caloric diet regime.
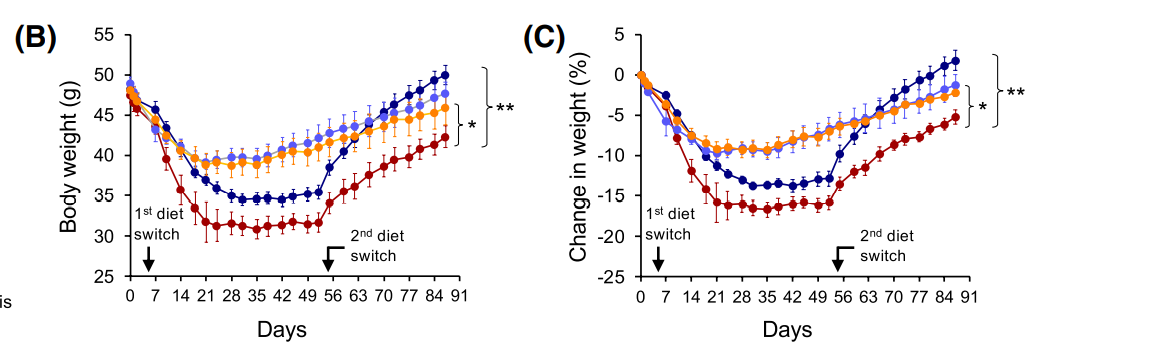
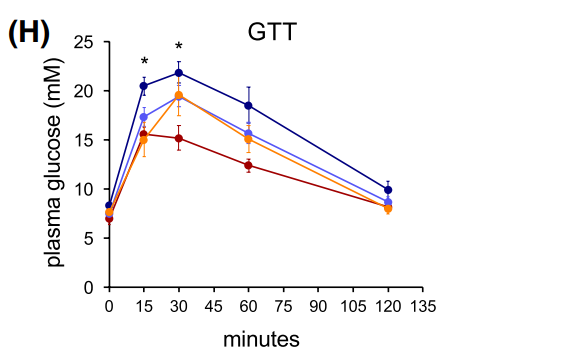
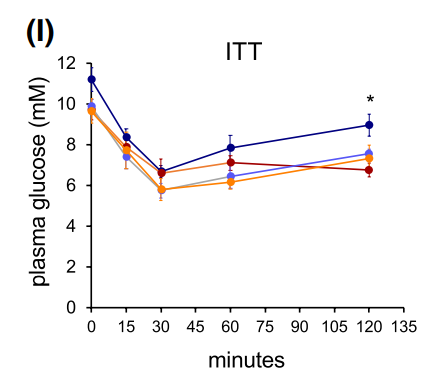
Figure 6 Body weights and results of Glucose tolerance test (GTT) (H) and Insulin tolerance test (ITT) of control and mutant mice after constant HFD or double-switch (SD→HFD).
The study also found that acute ablation of ALK7 in adipose tissue synergizes with salicylate to attenuate diet-induced obesity. While induced ALK7 deletion by tamoxifen injection, the lab treated one half of mutants and controls with salicylate at the same time. Initially, both groups on salicylate showed weight loss, but only mutant mice on salicylate could maintain reduced weight for several weeks, and ended up with the same weight after 9 weeks on HFD.
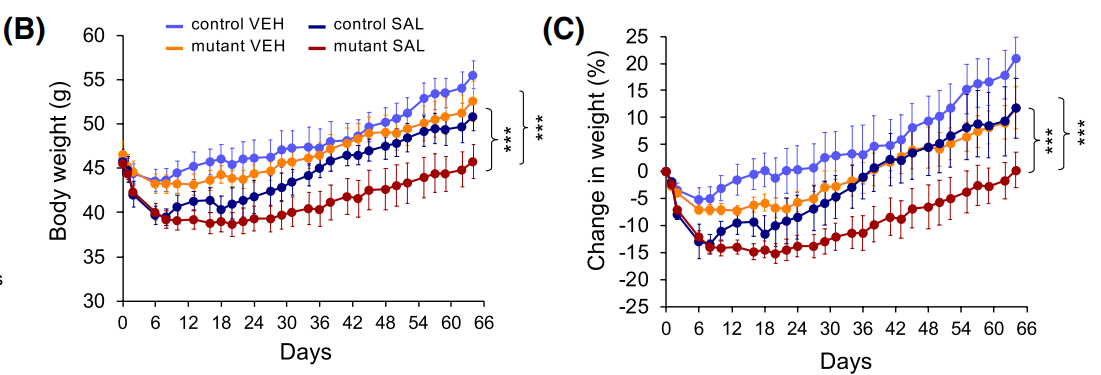
Figure 7 Body weights of obese control and mutant mice treated with salicylate (SAL) or vehicle (VEH).
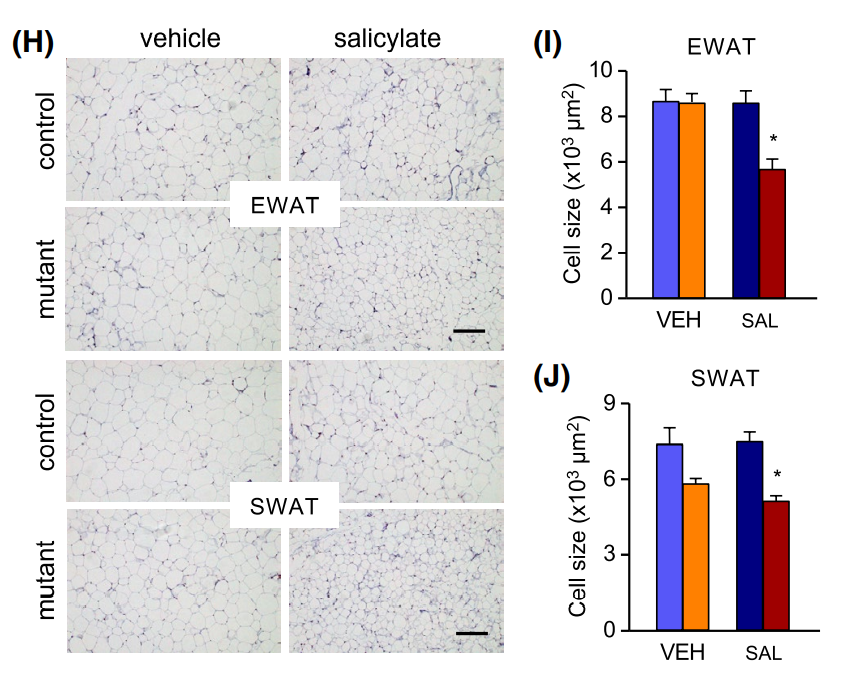
Figure 8 Adipocytes of obese control and mutant mice treated with salicylate (SAL) or vehicle (VEH).
This study has implications for the development of therapeutic strategies in human obesity and warrant efforts to identify safe and specific ALK7 inhibitors. In spite of existing limitations, blockade of ALK7, if achieved with low toxicity and minimal side effects, represents an important avenue to develop new strategies to combat human obesity and metabolic syndrome.
Raj Kamal Srivastava is the first author of this paper. Carlos F. Ibáñez serves as the corresponding author. Thank Goh Ket Yin for technical assistance and Meng Xie for comments on the manuscript. Thanks for the support from the Department of Biotechnology, India. This research was funded by grants from the Singapore National Medical Research Council, Aspiration Fund Partner from the National University of Singapore, and 2016-01538 from the Swedish Research Council.



