Novel Genetically Encoded Norepinephrine Fluorescent Sensors Published in “Neuron” by Dr. Yulong Li’s Group
On March 25, 2019, Yulong Li’s research group at Peking University in Beijing published the research article "A genetically encoded fluorescent sensor for rapid and specific in vivo detection of norepinephrine" online in Neuron. In this study, Li’s group described the successful development of the first genetically encoded norepinephrine fluorescent sensor, and subsequently used it to detect the dynamics of endogenous norepinephrine in live zebrafish and mice. This sensor specifically enables discrimination between structurally similar norepinephrine and dopamine with 1000-fold difference in affinity and will become a useful tool for studying norepinephrine-related neuronal circuits.
Norepinephrine (NE), an essential biogenic monoamine neurotransmitter, is involved in a wide range of physiological processes in the central and sympathetic nervous systems, including modulation of sensory information, regulation of attention, control of sleep and wake cycles, and learning and memory. Impaired NE secretion or signaling has been linked to a series of mental illnesses and neurodegenerative diseases. The ability to detect NE dynamics with high spatiotemporal resolution in a complex neural circuit is therefore especially critical to understand NE regulation and alterations in physiological or pathophysiological conditions. Unfortunately, currently available techniques are limited in terms of their sensitivity, specificity, spatiotemporal resolution and non-invasiveness, thus hampering our ability to understand NE dynamics in vivo.
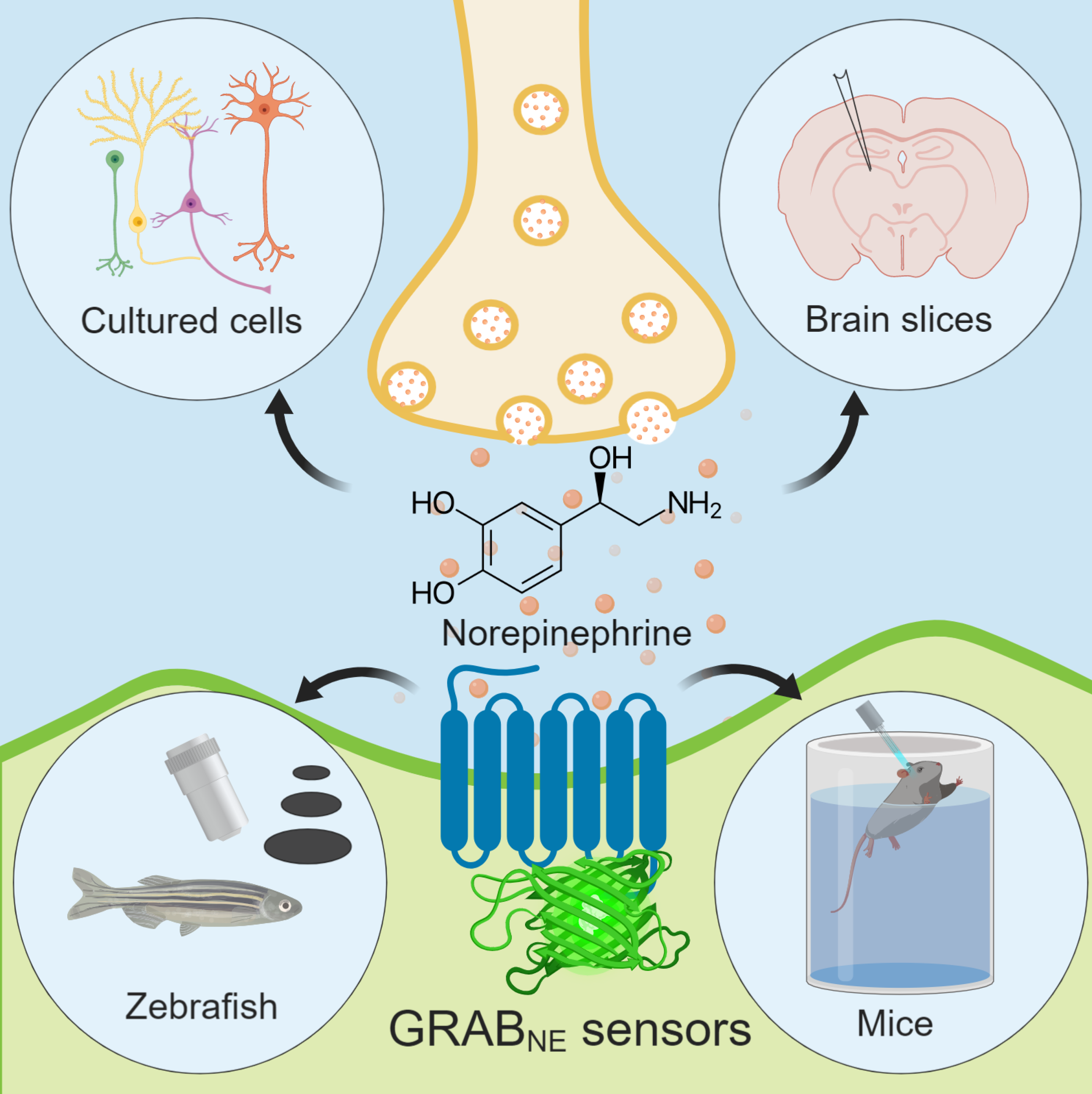
To overcome these technical obstacles, in this study, Yulong Li’s research group developed and optimized a family of G protein-coupled Receptor Activation Based NE (GRABNE) sensors. By inserting an environment-sensitive fluorescent protein (cpEGFP) at a specific location in the human adrenergic receptor, the conformational change induced by norepinephrine binding is converted into a fluorescent signal and, combined with existing imaging techniques, thus enables real-time monitoring of endogenous norepinephrine dynamics. After optimization, two versions of the GRABNE sensor were developed with EC50 values for NE of ~1 μM and ~100 nM (i.e., NE1m and NE1h, respectively), which are suitable for detecting synaptic or volume NE transmission with sensitivity, rapid kinetics, and high specificity.
G protein-coupled Receptor Activation Based NE (GRABNE) sensors
The genetically encoded GRABNE sensors can be introduced by transfection, virus vector injection, or genetic transformation to enable expression in cultured cells, brain slices, zebrafish, and mice. By generating a transgenic zebrafish line that pan-neuronally express GRABNE1m, the time-dependent specific release of NE induced by looming stimulation in the midbrain can be successfully detected. In addition, single-cell-resolution monitoring of NE release evoked by repeated visual stimuli can be achieved using sparsely labeled norepinephrine sensors. Using virus-mediated expression together with fiber photometry recording, optogenetic activation of LC-NE neurons was found to reliably induce increases in GRABNE1m fluorescence in freely moving mice. Similarly, a significant increase in GRABNE1m fluorescence was observed in the hypothalamus of freely moving mice during stress induced by forced swimming or tail suspension tests, suggesting modulation of NE dynamics during stress-associated behaviors.
Dr. Yulong Li, a Principal Investigator at the School of Life Sciences of Peking University, Peking University-Tsinghua Joint Center for Life Sciences, PKU-IDG/McGovern Institute for Brain Research, is the corresponding author of this paper. Jiesi Feng, in the Li research group, serves as the first author; Miao Jing from CIBR contributed to this study. Collaborators of this work include: Jiulin Du’s research group at the Shanghai Institute of Neuroscience of the Chinese Academy of Sciences; Dayu Lin’s research group at New York University; Guohong Cui’s research group at the National Institutes of Health; J. Julius Zhu’s research group at the University of Virginia School of Medicine; and S. Andrew Hires research group at the University of Southern California. This work was supported by the State Key Laboratory of Membrane Biology at Peking University School of Life Sciences, Peking University-Tsinghua Joint Center for Life Sciences, National Basic Research Program of China (973 Program), the General Program of National Natural Science Foundation of China, and the NIH BRAIN Initiative grant.



