Neuron: Minmin Luo's Team Discovers the Neural Mechanisms of Reward Devaluation and Its Regulation in Depression and Obesity
Rewards are essential for the survival and reproduction of humans and animals, such as food, water, and sex. They can motivate individuals to seek behaviors that satisfy basic needs and generate pleasurable feelings, while guiding learning and memory formation. The value of rewards is not fixed, but dynamically changes, influenced by an individual's physiological needs and the history of reward acquisition. In other words, the same reward may have different effects at different times and contexts. For example, delicious food and beautiful music are common pleasures in our lives, but we often find that repeated consumption of the same food or listening to the same song too many times can make these enjoyable stimuli become increasingly bland. The process of obtaining rewards can lead to a decrease in subjective pleasure, making us feel satiated and bored. This phenomenon is known as sensory-specific satiety, and the resulting decrease in subjective reward value is also called reward devaluation. The process of reward devaluation allows individuals to dynamically adjust their reward intake and stop consuming a particular reward at the appropriate time, promoting the search for new rewards. Maintaining an appropriate sensitivity to reward devaluation is crucial for our survival and life. Abnormal changes in this sensitivity may lead to related mental disorders. For example, insensitivity to reward devaluation is associated with binge eating and obesity, while excessive sensitivity to devaluation can reduce motivation to seek rewards, leading to anhedonia (the core symptom of depression).
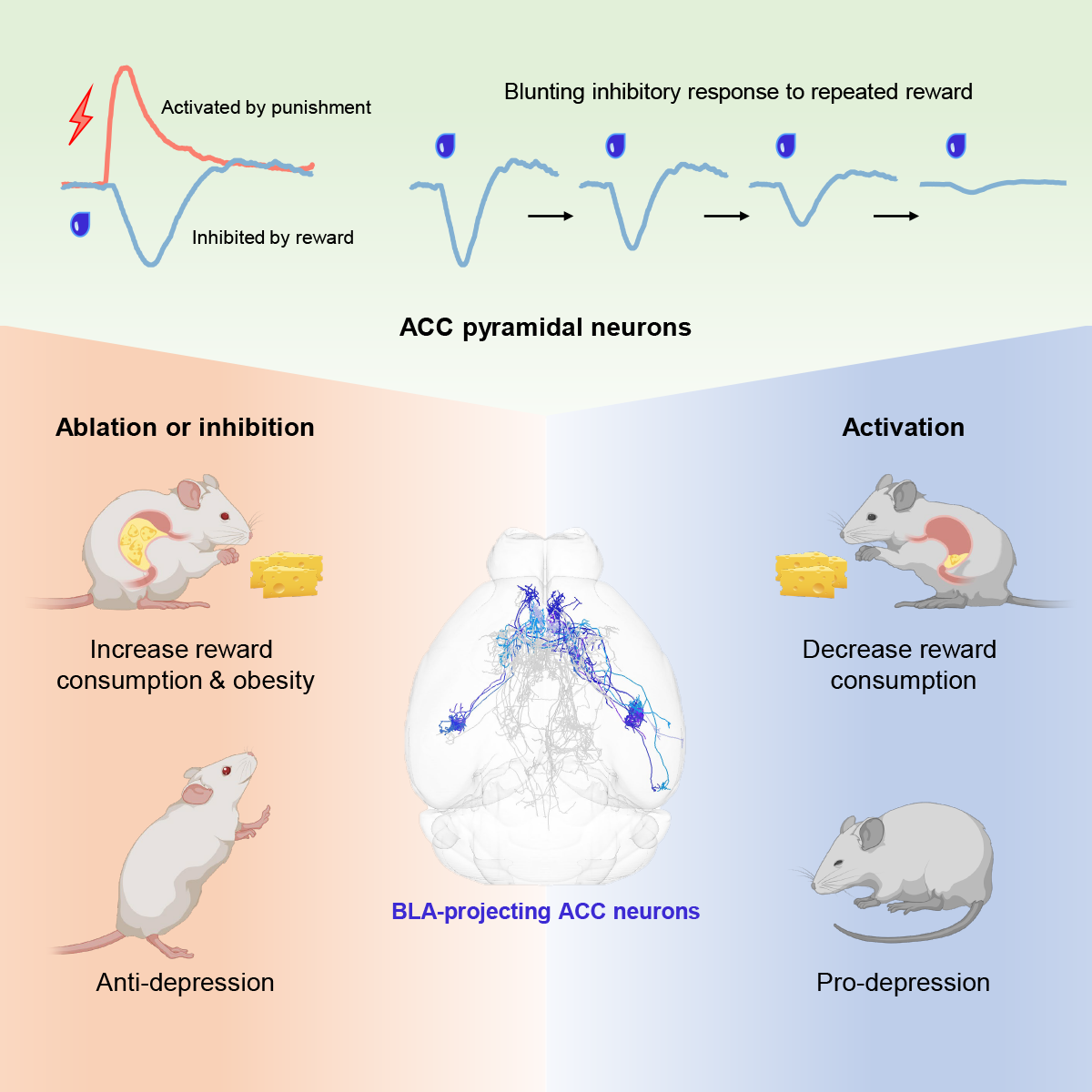
Although the phenomenon of sensory-specific satiety has been observed in various animal models, including humans and mice, the central neural mechanisms underlying it are poorly understood, and its impact on human health and mental illness remains unexplored. On September 20th, 2023, Minmin Luo's laboratory at the Chinese Institute for Brain Research (CIBR) published a research article in Neuron entitled "A Corticoamygdalar Pathway Controls Reward Devaluation and Depression Using Dynamic Inhibition Code" revealing that pyramidal neurons in the anterior cingulate cortex (ACC), especially a subtype projecting to the basolateral amygdala (BLA), can encode and regulate reward devaluation. Manipulating these neurons can adjust the sensitivity of mice to reward devaluation, leading to overconsumption or underconsumption of rewards and the development of obesity or depression-like states. Conversely, this manipulation can be used to attempt to treat related diseases such as depression.
The research team first recorded the responses of pyramidal neurons in the ACC to different rewards and punishments at both the population and single-cell levels using fiber photometry and miniscope imaging (Figure 1). Regardless of whether the mice actively consumed rewards (thirsty mice licking water bottles or hungry mice eating high-fat food) or passively received liquid rewards delivered from cheek fistula, pyramidal neurons in the ACC were significantly inhibited, and the strength of this inhibition was positively correlated with reward value. In contrast, different punishing stimuli (bitter water, electric shocks) activated these neurons. The results of miniscope imaging showed that 41% of pyramidal neurons in the ACC were inhibited by rewards, 21% were activated by rewards, and the rest showed no significant response, indicating an overall inhibitory response to rewards.
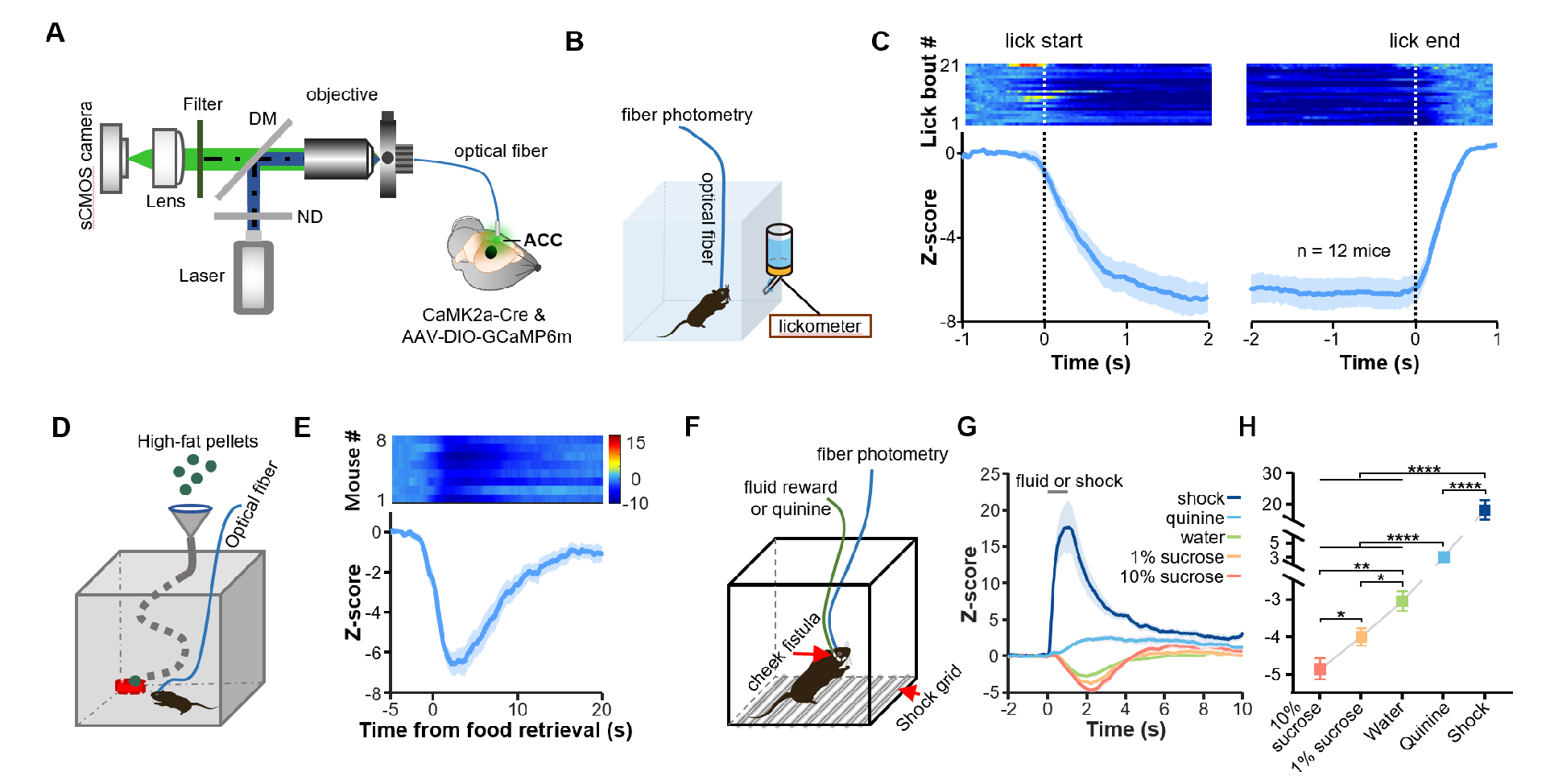
Figure 1: ACC pyramidal neurons exhibit overall inhibitory responses to rewards.
Based on these recorded results, the researchers then investigated whether ablating, inhibiting, or activating these neurons would affect reward intake (Figure 2). In a water reward intake test, thirsty mice were only allowed to access and drink from a water bottle during a 30-minute test period each day, which lasted for 4-5 days. Using kainic acid (KA) excitotoxicity to ablate ACC neurons or inducing apoptosis of pyramidal neurons in the ACC using caspase3 expression, thirsty mice showed excessive water reward intake, and in extreme cases, mice even died because they could not stop drinking water rewards in time. Temporarily reversible inhibition or activation of pyramidal neurons in the ACC using chemogenetics significantly increased or decreased water reward intake, respectively. When high-fat food which is a strong rewarding stimulus was provided to mice for a long time, mice with pyramidal neurons ablation in the ACC consumed more high-fat food and rapidly gained weight, becoming very obese. Interestingly, damaging or inhibiting pyramidal neurons in the ACC only increased reward intake. If the mice were not thirsty, inhibiting pyramidal neurons in the ACC did not prompt them to drink water voluntarily. This also suggests that inhibiting pyramidal neurons in the ACC do not make mice thirsty or directly induce motivation to consume rewards.
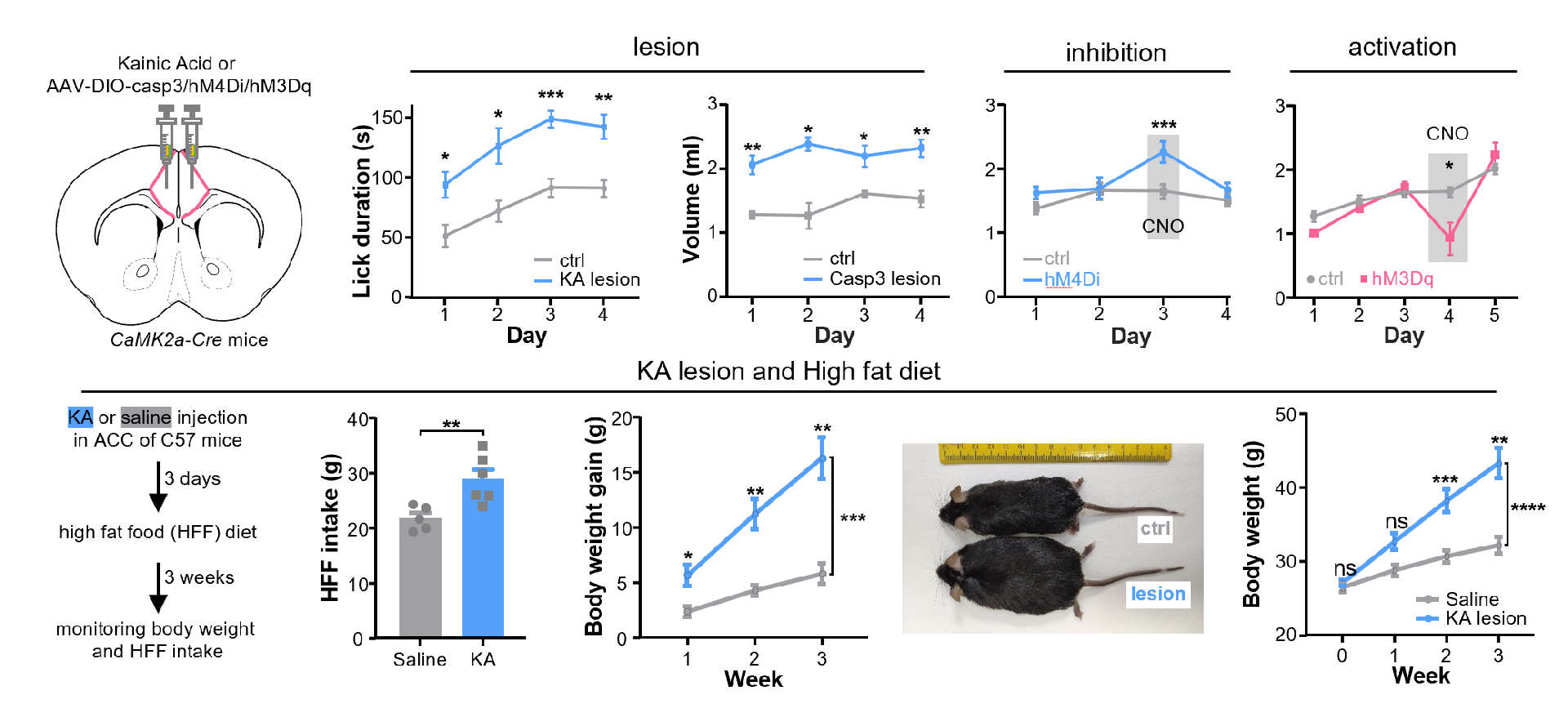
Figure 2: ACC pyramidal neurons suppress reward consumption.
Repeated intake of the same reward leads to its devaluation, and the pleasure it brings gradually decreases. To explore how pyramidal neurons in the ACC encode this dynamic process, the researchers directly pumped water or sugar water rewards into the oral cavities of mice through a cheek fistula, repeating the experiment for 100 trials, and the passive receipt of rewards ensured that the reward amount of each trial was the same (Figure 3). The results of fiber photometry recording showed that at the population level, pyramidal neurons in the ACC were strongly inhibited by rewards at the beginning, and this inhibition gradually weakened with repeated delivery of rewards. By averaging the 100 trials into 5 blocks for statistical analysis, a significant reduction in reward-induced inhibition was observed. At the same time, depression model mice (CRS mice) showed a faster reduction, suggesting that reward devaluation in depressed mice is faster. Two-photon microscopy imaging recordings revealed that at the single-cell level, the number of pyramidal neurons in the ACC inhibited by rewards gradually decreased with repeated delivery of rewards, while the number of neurons activated by rewards did not change significantly.
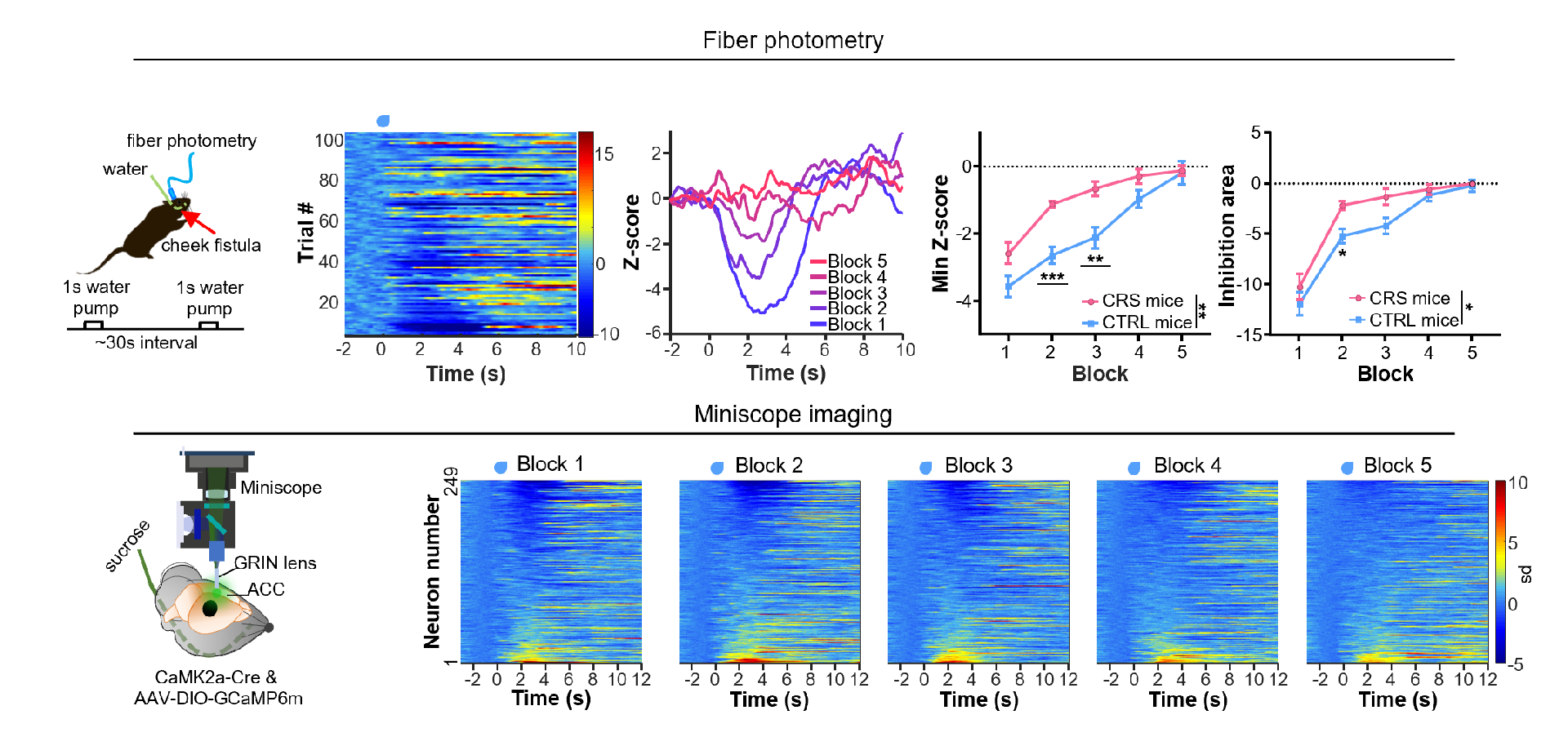
Figure 3: ACC pyramidal neurons encode reward devaluation by blunting reward-induced inhibition.
To further explore the relationship between the ACC and reward devaluation, the researchers improved the behavioral paradigm used in other animal models to test reward devaluation (Figure 4). After mice consumed a sufficient amount of a certain reward, they would reduce their preference for that reward and tend to choose another reward. When pyramidal neurons in the ACC were ablated or inhibited, mice did not reduce their preference for the previous reward, indicating that normal activity of pyramidal neurons in the ACC is necessary for reward devaluation. Depression animal models showed hypersensitivity to reward devaluation, and this defect could be alleviated by the fast-acting antidepressant ketamine. Chemogenetic inhibition of pyramidal neurons in the ACC could also alleviate the reward devaluation feature of depression-like animals' hypersensitivity, as well as alleviate depression-like behaviors such as anhedonia and behavioral despair.
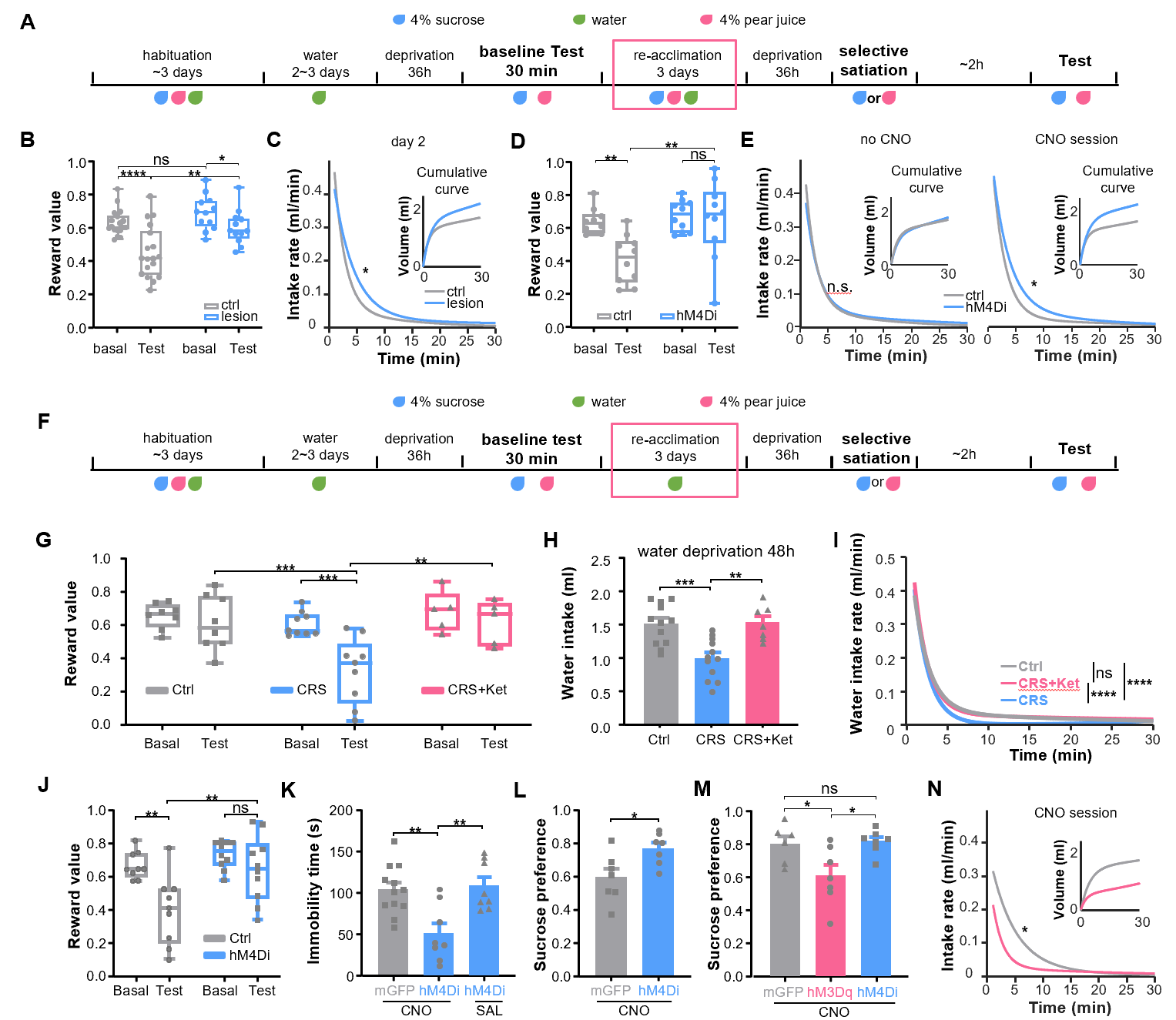
Figure 4: ACC pyramidal neurons control reward devaluation and depression-like behaviors.
ACC pyramidal neurons project axonal terminals into numerous downstream brain areas. To investigated which ACC projection(s) might function in controlling reward intake and devaluation, researchers reconstructed the complete morphology of individual neurons and identified five distinct subpopulations of ACC neurons that project to the VTA, BLA, NAc, MS, and OFC (Figure 5). Further tracing experiments confirmed that these five subpopulations of neurons have different collateral patterns and specific distributions in cell body locations.
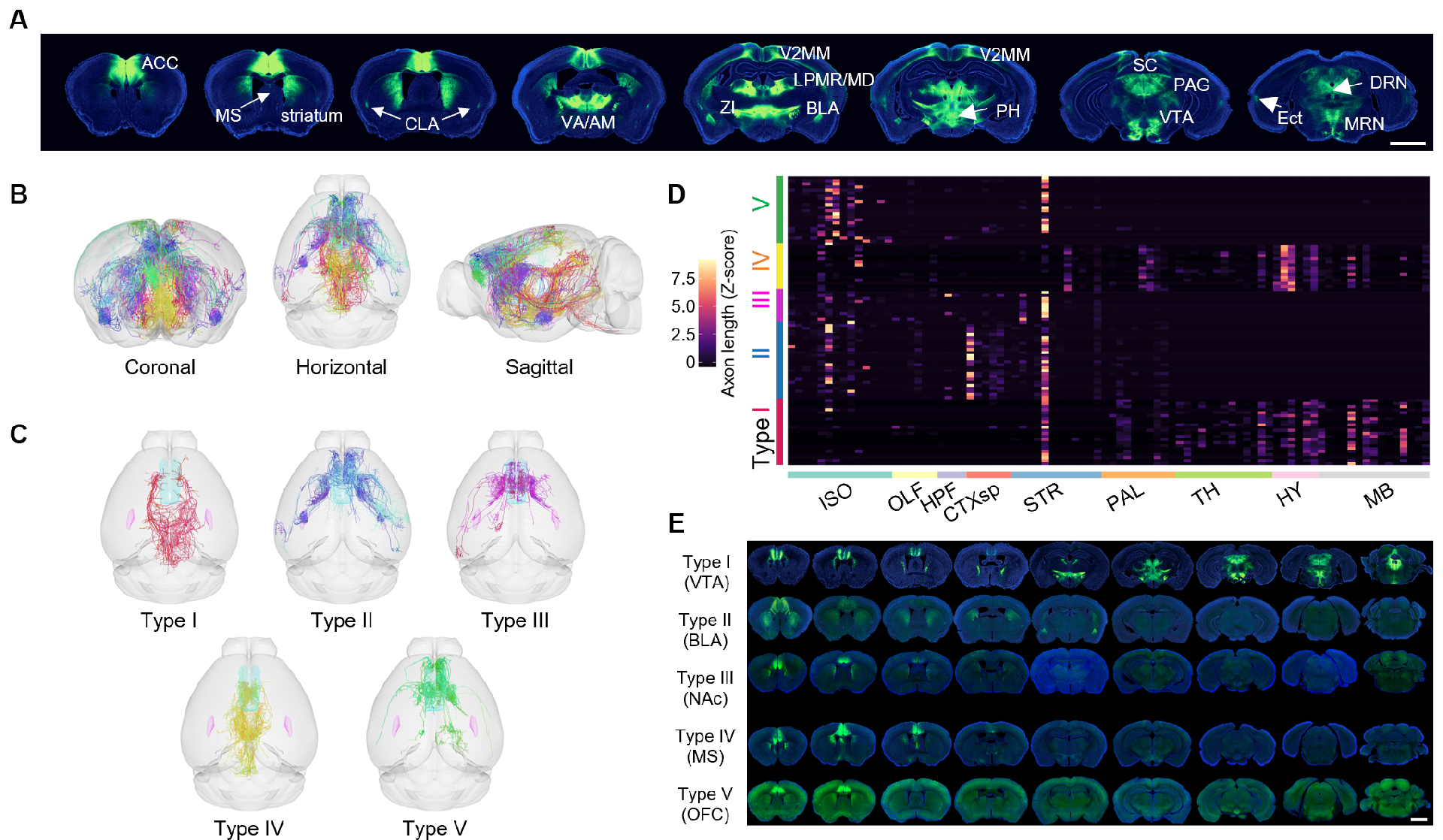
Figure 5: ACC pyramidal neurons comprise multiple subtypes with distinct projection patterns.
To verify which subpopulations of neurons can control reward devaluation and depression-like behavior, researchers conducted extensive behavioral screening tests (Figure 6). By retrogradely labeling ACC neurons projecting to 11 downstream brain regions, including the five representative brain regions mentioned above, researchers selectively ablated each subpopulation of neurons and conducted water reward intake tests. Only ablating the basolateral amygdala (BLA)-projecting ACC neurons significantly increased reward intake and significantly abolished reward devaluation. Fiber photometry and miniscope imaging also confirmed that this subpopulation of neurons could encode reward devaluation by gradually weakening inhibitory signals. Activation of this subpopulation of neurons sensitized reward devaluation, reduced reward intake, and induced depression-like behavior; while ablating this subpopulation of neurons in depressed animals alleviated depression-like symptoms.
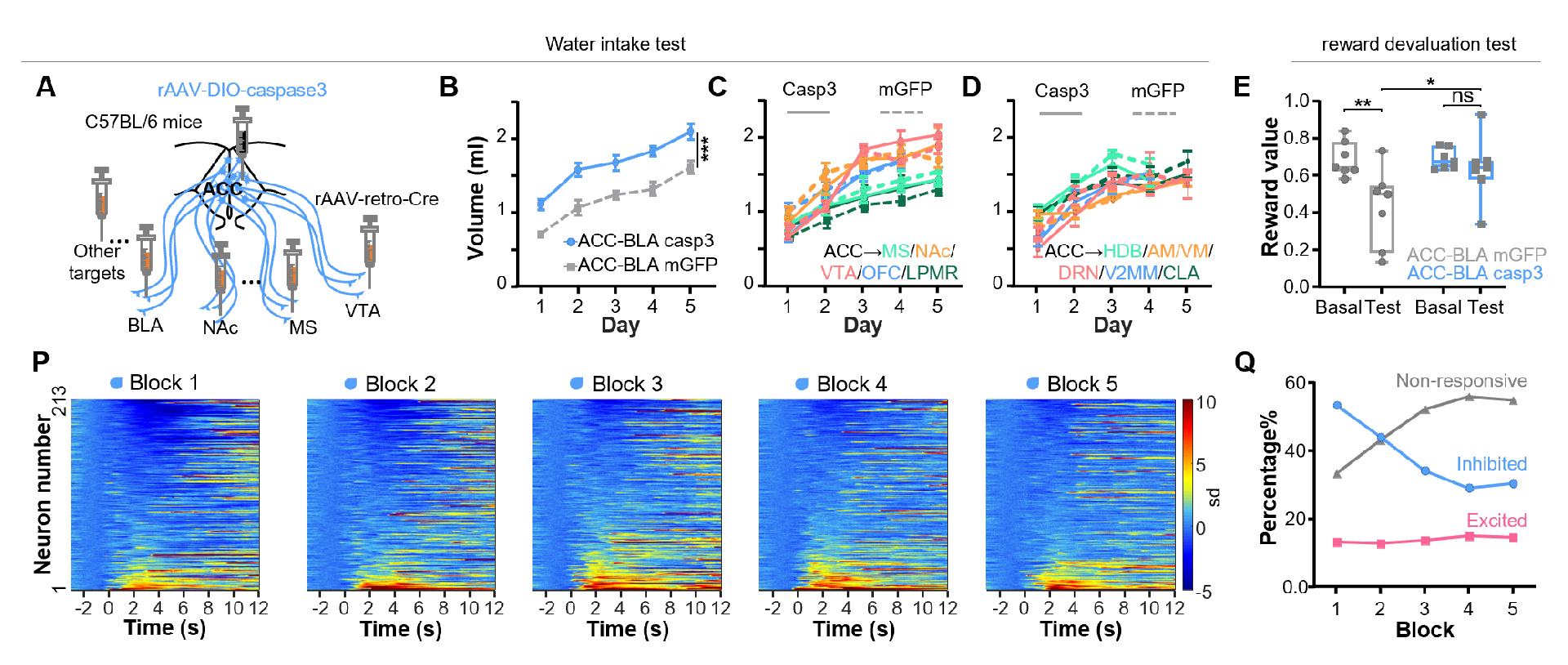
Figure 6: BLA-projecting ACC neurons regulate reward devaluation and depressive behaviors.
In summary, this study found that the pyramidal neurons in the anterior cingulate cortex, especially the subtype projecting to the basolateral amygdala, can encode and control reward devaluation. Ablating or inhibiting these neurons blunt reward devaluation, causing animals to consume excessive rewards, which can lead to extreme conditions such as obesity or water intoxication. On the other hand, activating these neurons can sensitize reward devaluation, reduce reward intake, and exhibit depressive behaviors. This study differs from common findings that a nucleus can "statically" encode reward or aversion. Instead, it focuses on the anterior cingulate cortex, an important brain cortical area, in the "dynamic" changes of reward value. This discovery helps increase understanding and explanation of depression-like behaviors, including anhedonia, and proposes new treatment strategies for depression and eating disorder-related diseases.
Article related information:
Dr. Zhengwei Yuan, a postdoctoral fellow in the laboratory of Dr. Minmin Luo at the Chinese Institute for Brain Research, and Dr. Zhongyang Qi, a jointly trained Ph.D. student in the Luo lab and Huazhong University of Science and Technology, are co-first authors of the paper. Dr. Minmin Luo is the corresponding author. Other authors of this paper include Dr. Ruiyu Wang, Dr. Yuting Cui, Dr. Qiru Feng, Dr. Rui Lin, Ruicheng Dai, Guoli Wu from the National Institute of Biological Sciences (Beijing) and Chinese Institute for Brain Research (Beijing), as well as Professor Ling Fu, Dr. Sile An, Dr. Anan Li, Professor Hui Gong, and Professor Qingming Luo from Huazhong University of Science and Technology. This study was supported by China Brain Initiative Grant (STI2030-Major Projects 2021ZD0202803), Research Unit of Medical Neurobiology at Chinese Academy of Medical Sciences (2019RU003), New Cornerstone Investigator Program, and Beijing Municipal Government.



