Nature | Adenosine Signalling Drives Antidepressant Actions of Ketamine and ECT | Minmin Luo Lab
Depression poses a major challenge in clinical psychiatry. Ketamine and electroconvulsive therapy (ECT) are among the very few interventions known to produce rapid and sustained antidepressant effects. However, their use is limited by significant drawbacks: ketamine can cause dissociative side effects and has an addictive potential, while ECT may lead to short-term memory loss and cognitive impairments. The long-elusive common neurobiological mechanism shared by these two distinct treatments has been a critical barrier to developing safer and more effective therapies.
Recently, the laboratory of Dr. Minmin Luo at the Chinese Institute for Brain Research, Beijing (CIBR), published a study in Nature titled "Adenosine Signalling Drives Antidepressant Actions of Ketamine and ECT". The research identifies a signaling molecule in the brain called adenosine as the central pathway through which both ketamine and ECT take effect. Based on this discovery, the team also proposed an "Acute Intermittent Hypoxia Intervention"(aIH), which safely and controllably triggers adenosine release in the brain, achieving significant antidepressant outcomes.
The researchers first used a genetically encoded adenosine sensor (GRABado) combined with fiber photometry to monitor real-time dynamics of extracellular adenosine concentration in specific brain regions of mice. They found that after administering a sub-anesthetic dose of ketamine, a rapid and sustained increase in adenosine levels occurred in the medial prefrontal cortex (mPFC) and hippocampus, but not in the nucleus accumbens. Similarly, ECT also induced comparable adenosine release in the mPFC.
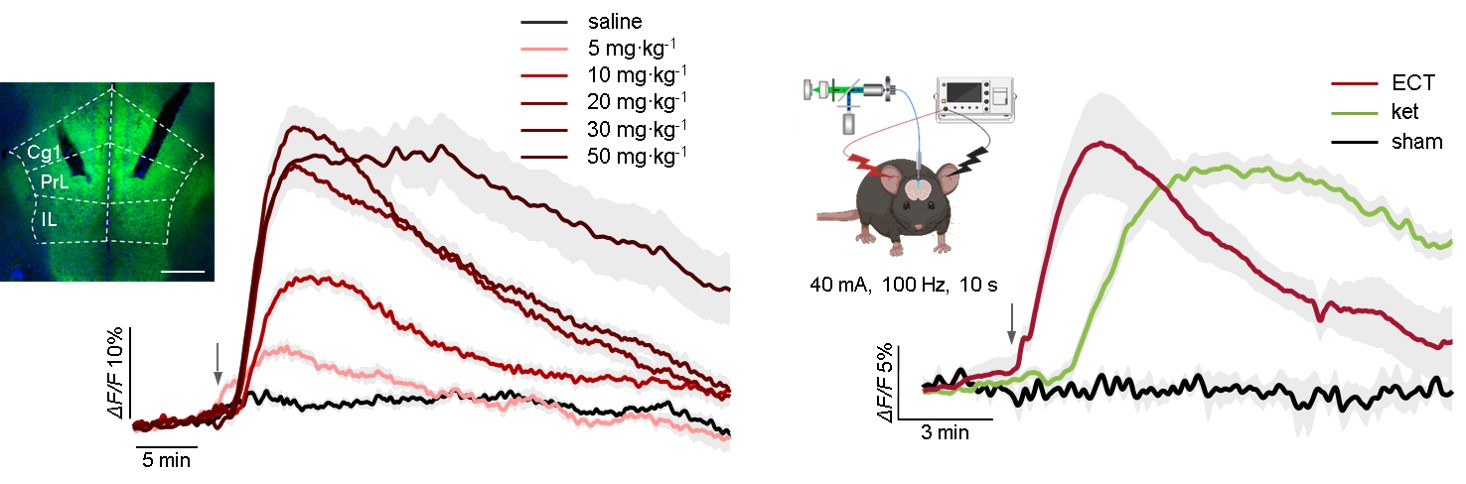
Figure 1: Ketamine and ECT induce a surge of adenosine in the medial prefrontal cortex.
Subsequently, the researchers confirmed the necessity of adenosine signaling for the antidepressant effects using genetic and pharmacological approaches. In mice genetically lacking either the A₁ or A₂A adenosine receptors, the antidepressant effects of both ketamine and ECT, as measured by behavioral tests like the forced swim test and sucrose preference test, were completely abolished. In wild-type mice, systemic administration of A₁ or A₂A receptor antagonists also blocked ketamine's antidepressant effects. Importantly, the genetic deletion of the receptors did not affect ketamine-induced adenosine release itself, nor did it alter the hyperlocomotion induced by ketamine, indicating that the adenosine pathway specifically mediates the therapeutic action rather than non-specific behavioral effects.
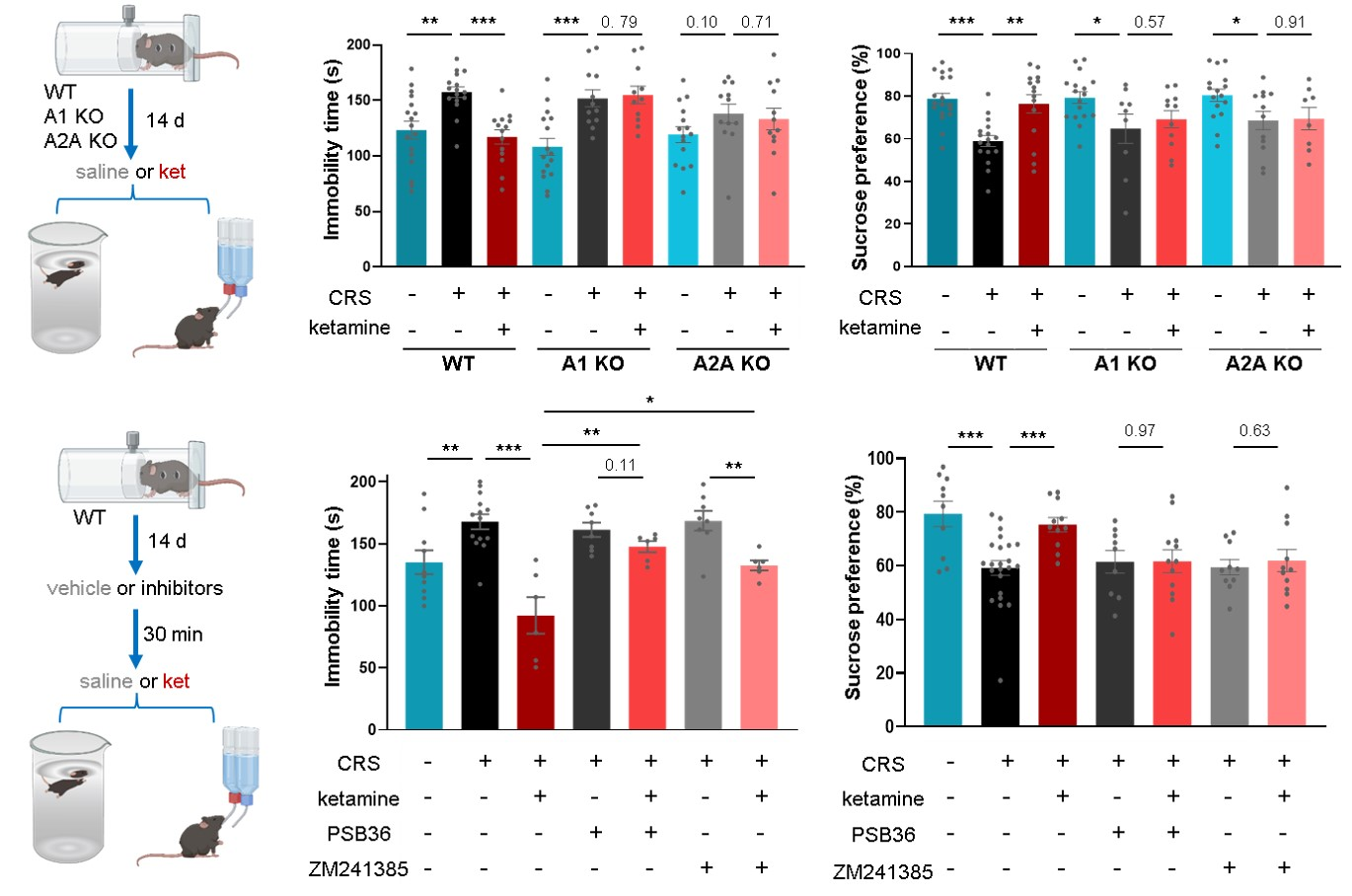
Figure 2: Genetic and pharmacological evidence demonstrates that adenosine signaling mediates the rapid antidepressant phenotype of ketamine.
To pinpoint the brain region where adenosine acts, the team conducted functional validation at the circuit level. They found that directly injecting adenosine into the mPFC, or using optogenetics to specifically activate astrocytes in this region to induce endogenous adenosine release, was sufficient to produce rapid antidepressant behavioral effects in depressed mice. Conversely, using AAV-mediated CRISPR/Cas9 technology to specifically knock down A₁ or A₂A receptors in the mPFC blocked the therapeutic effect of systemically administered ketamine. These results collectively indicate that the mPFC is a critical node where adenosine signaling mediates rapid antidepressant action.
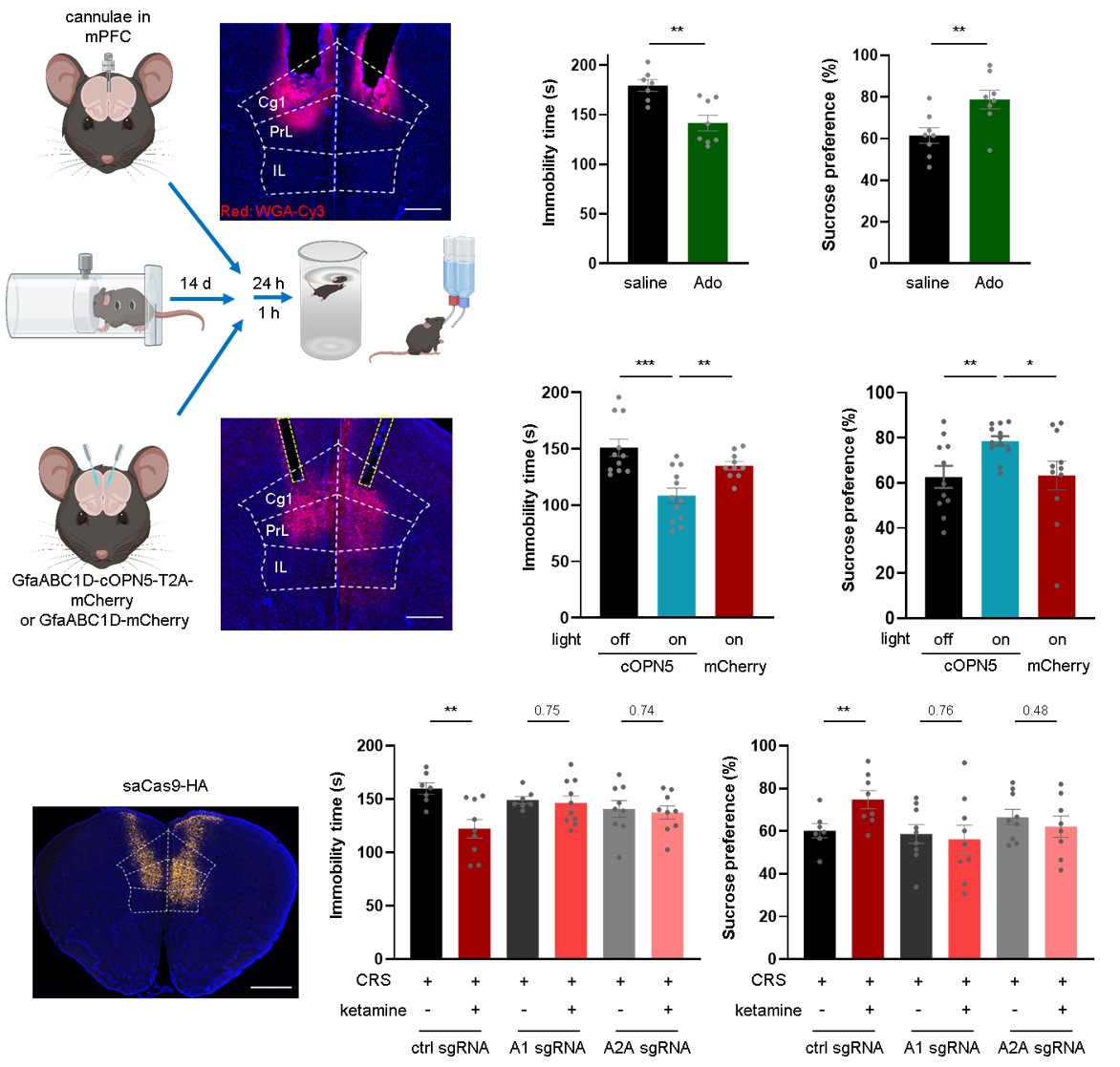
Figure 3: Evidence for the sufficiency and necessity of mPFC adenosine signaling for the antidepressant phenotype.
Mechanistically, the study delved deeper into the source of the increased extracellular adenosine levels induced by ketamine. The researchers found that ketamine did not increase extracellular ATP/ADP levels, and its effect persisted in CD73 knockout mice, ruling out extracellular nucleotide hydrolysis as the primary source. Further experiments showed that inhibiting equilibrative nucleoside transporters (ENT1/2) significantly attenuated ketamine-induced adenosine release, suggesting an intracellular origin. Using the ATP/ADP ratio sensor PercevalHR, the researchers observed that ketamine rapidly decreased the ATP/ADP ratio within excitatory neurons, inhibitory neurons, and astrocytes in the mPFC. This metabolic change occurred before the rise in extracellular adenosine. In vitro mitochondrial metabolic flux analysis revealed that ketamine, at therapeutically relevant concentrations, directly inhibited the metabolic flux of the mitochondrial tricarboxylic acid (TCA) cycle. Together, this evidence outlines a novel pathway: ketamine modulates cellular energy metabolism, increasing intracellular adenosine, which is then released into the extracellular space via nucleoside transporters.
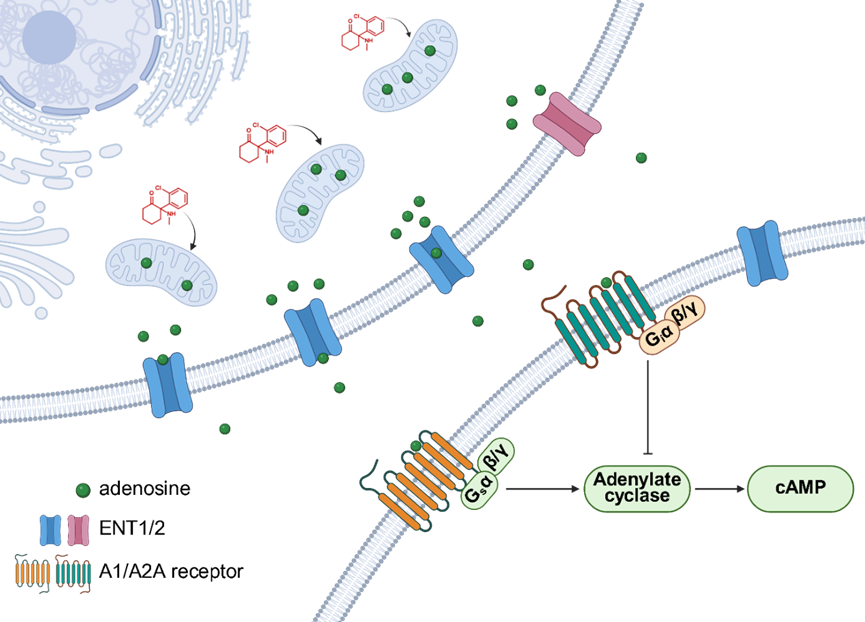
Figure 4: Mechanism of ketamine-induced adenosine release in the brain.
Building on this mechanistic understanding, the researchers designed and screened a series of ketamine derivatives using "enhanced adenosine release" as a key phenotypic indicator. Among them, deschloroketamine (DCK), even at lower doses, induced a stronger and more prolonged adenosine signal and demonstrated superior antidepressant efficacy in behavioral tests compared to ketamine, while causing significantly less hyperlocomotion, a proxy for dissociative side effects. Notably, a compound's ability to release adenosine did not significantly correlate with its potency for inhibiting NMDA receptors, suggesting a novel mechanism of antidepressant action independent of NMDAR inhibition.
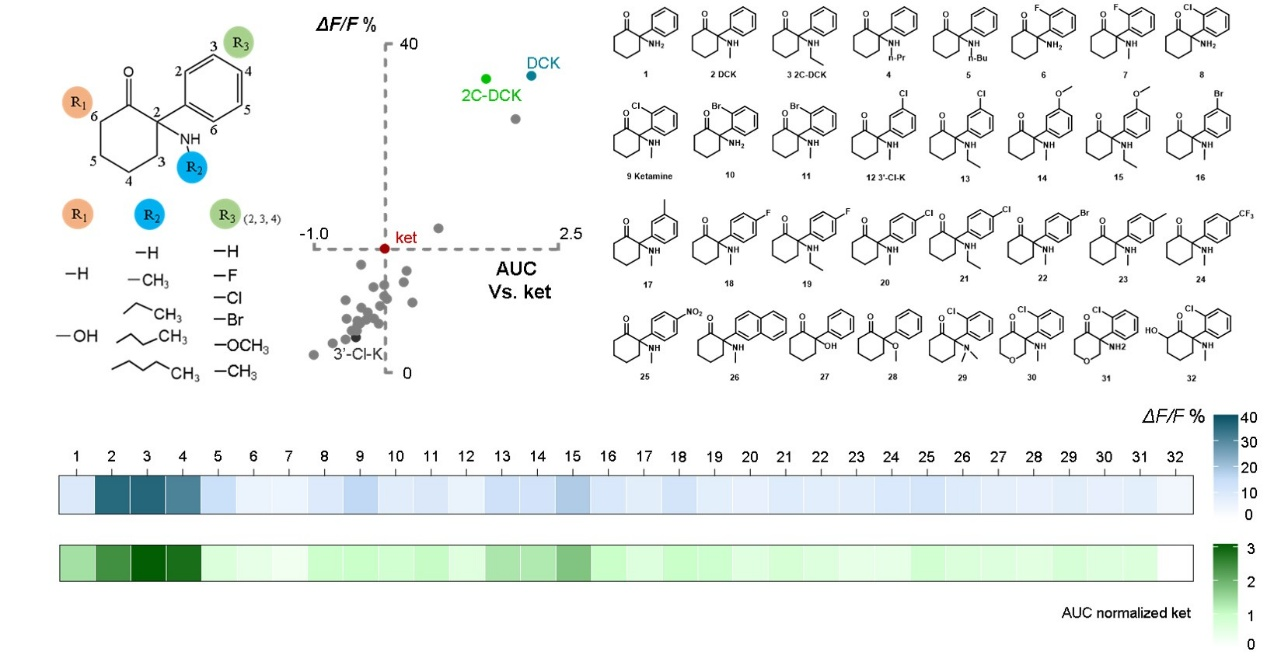
Figure 5: Adenosine release-based drug screening system.
Furthermore, the researchers explored the therapeutic potential of non-pharmacological methods to elevate brain adenosine levels. They found that a controlled acute intermittent hypoxia (aIH) intervention could safely trigger adenosine release in the mPFC and produce rapid antidepressant effects in depressed mice, which were dependent on both A₁ and A₂A receptors.
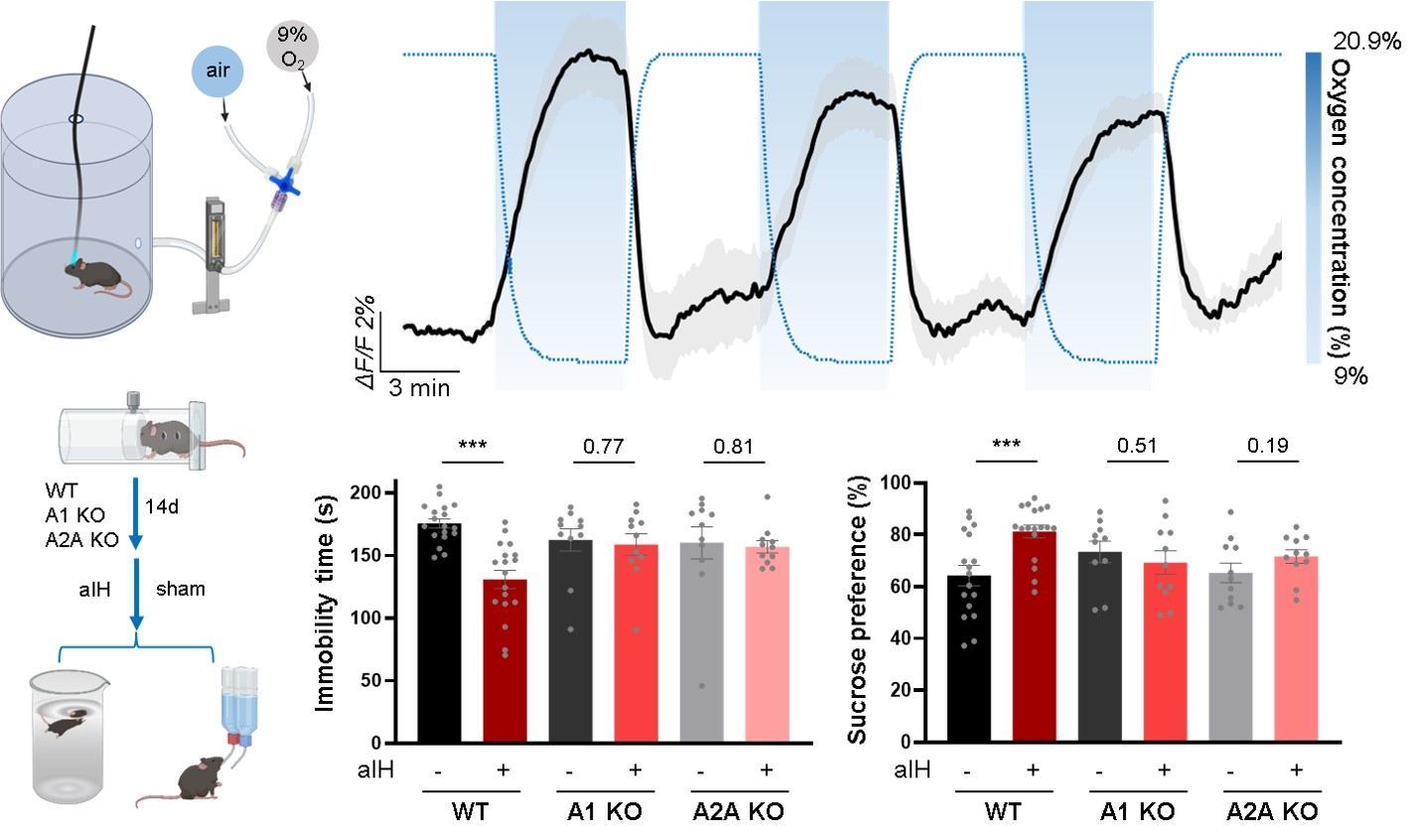
Figure 6: Acute Intermittent Hypoxia induces adenosine release and alleviates depression-like behaviors.
In summary, this study identifies and confirms the adenosine signaling pathway as the common critical mechanism underlying the rapid antidepressant effects of both ketamine and ECT. This discovery not only deepens our understanding of how rapid antidepressants work but also provides a solid theoretical foundation and a clear target for developing a new generation of antidepressant strategies—including both small-molecule drugs and non-pharmacological interventions—based on the regulation of adenosine signaling, with the potential for fewer side effects.
This study was led by Dr. Minmin Luo, with two co-first authors Chenyu Yue and Na Wang from the Chinese Institute for Brain Research, Beijing (CIBR) & the Chinese Academy of Medical Sciences conducting the key behavioral and fiber photometry experiments.
The project represents a significant multi-disciplinary collaboration. The primary research was conducted at CIBR, under the supervision of senior author Minmin Luo. Key scientific collaborations were essential to the study's success:
• Chemical Synthesis: The novel ketamine analogues were synthesized by a team led by Xiaohui Wang at the Changchun Institute of Applied Chemistry, Chinese Academy of Sciences (CIAC).
• Essential Biological Tools: The critical, genetically encoded GRAB sensor AAV vectors were provided by experts Yulong Li from Peking University and Zhaofa Wu from the Institute of Genetics and Developmental Biology, CAS.
• Metabolomics: The metabolomic analysis was conducted by Huijie Mi, with data interpretation advised by Wooping Ge at CIBR.
The research was supported by a broad range of major national and municipal funding bodies in China. Significant support was provided by the New Cornerstone Investigator Program, the CAMS Innovation Fund for Medical Sciences, the China Brain Initiative, and the National Natural Science Foundation of China, with several key grants awarded to the senior authors, including Minmin Luo and Yulong Li.



