
Yang Shi
2012-2018 Washington University in St. Louis, Ph.D., Neuroscience
2007-2011 China Agricultural University, B.S., Biological Sciences
2022-now Assistant Investigator, Chinese Institute for Brain Research, Beijing
2019.1-2022.6 Postdoc Associate, Washington University in St. Louis
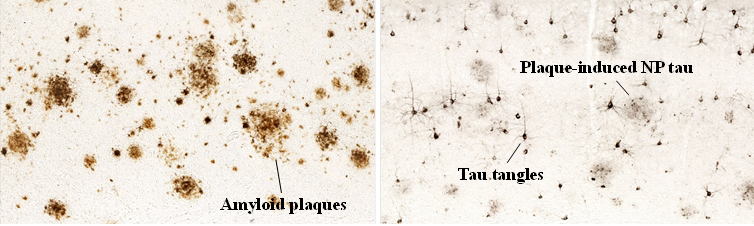
Alzheimer’s disease (AD) is the No.1 cause of dementia, and one of the most challenging unsolved medical problem in the world. AD patients show severe neurodegeneration and cognitive impairment. Pathologically, AD is characterized by extracellular deposition of Aβ plaques and intraneuronal accumulation of neurofibrillary tangles (see pictures below). Aβ plaques begin to accumulate in patients’ brain 10-20 years prior to their symptom onset. When the plaque load reaches plateau, tau pathology arises, followed by onset of neurodegeneration and cognitive decline. The cause of late-onset AD is still unclear. In the past century, the mainstream opinion deems Aβ as the key player that triggers the downstream detrimental cascades, and APOE, the strongest genetic risk factor for late-onset AD, was believed to affect AD primarily by regulating the amyloid pathology. However, therapeutic strategies targeting Aβ have not been successful so far.
Recently, accumulating evidence indicates that the immune system plays an essential role in regulating AD pathogenesis. We previously found in a tauopathy mouse model that microglia, the immune cell type in the brain, serve as the primary driving force of both tau pathogenesis and neurodegeneration (Shi et al, JEM, 2019). When microglia are absent, the formation of the pathological tau literally stops, and neurodegeneration is almost fully prevented. APOE as a lipid-binding protein, has potent immunomodulatory functions by itself, and can directly regulate tau pathogenesis and tau-associated neurodegeneration by controlling microglial activation, the process of which is independent of the impact of APOE on amyloid pathology (Shi et al, Nature, 2017).
While microglia can directly regulate neurodegeneration in the brain, our body’s immune system is functionally connected, and the peripheral immune system likely also participates in regulating AD pathogenesis via crosstalking with microglia or other CNS cell types. Although the brain was traditionally deemed as an immune-privileged organ, early and recent studies provide convincing evidence of brain infiltration by peripheral immune cell types in the normal brain, and the cell number is increased in AD. In addition, the composition and activation status of peripheral immune cell types in the blood and cerebrospinal fluid of AD patients are also altered. This evidence suggests potential involvement of the peripheral immune system in regulating AD.
My lab utilizes multidisciplinary approaches to systematically study the role of the neuro-immune axis in regulating Alzheimer’s disease, and to develop novel therapies based on the neuro-immune interaction. We are interested in multiple research directions:
1. The mechanisms underlying APOE’s immune regulatory function
2. The role of the peripheral immune system in regulating AD
3. Therapy development targeting microglia and the immune system
4. Neuro-immune crosstalk
Whichever research background you come from, if you are interested in learning the cool science behind Alzheimer’s disease and the neuro-immune axis, or hope to make your own contribution to curing Alzheimer’s disease, don’t hesitate to contact me at shiyang@cibr.ac.cn. We are a multidisciplinary research lab that welcome the bright minds from all fields to brainstorm together!

2021 Rainwater Foundation Tau Leadership Award
2020 Marian Kies Award of American Society for Neurochemistry
2019 Washington University Neuroscience Tom Thach Award
2019 The 144th Annual Meeting of the American Neurological Association Travel Award
2018 David M. Kipnis Award in Biomedical Sciences
2017 Rainwater Foundation Tau consortium fellowship
2016 Washington University Neuroscience Tom Thach Award
1. TCW, J., Qian, L., Pipalia, N., Chao, M., Liang, S., Shi, Y., Jain, B. R., Bertelsen, S. E., Kapoor, M., Marcora, E., Sikora, E., Andrews, E. J., Martini, A. C., Karch, C. M., Head, E., Holtzman, D. M., Zhang, B., Wang, M., Maxfield, F. R., Poon, W. W., Goate, A. M. Cholesterol and matrisome pathways dysregulated in APOE ε4 human microglia and astrocytes. Cell, Accepted, (2022)
2. Shi, Y., Andhey, P.S., Ising, C., Wang K., Snipes, L.L., Boyer, K., Lawson. S., Yamada, K., Qin, W., Manis, M., Remolina J., Benitez, B.A., Schmidt, R.E., Artyomov, M., Ulrich, J., Holtzman, D.M. Overexpressing low-density lipoprotein receptor reduces tau-associated neurodegeneration in relation to apoE-linked mechanisms. Neuron (2021).
Neuron Preview: Mendiola, A. S., Tognatta, R., Yan, Z., Akassoglou, K. ApoE and immunity in Alzheimer’s disease and related tauopathies: Low-density lipoprotein receptor to the rescue (2021).
3. Wang, C., Xiong, M., Gratuze, M., Bao, X., Shi, Y., Andhey, P.S., Manis, M., Schroeder, C., Yin, Z., Madore, C., Butovsky, O., Artyomov, M., Ulrich, J., Holtzman, D.M. Selective removal of astrocytic APOE4 strongly protects against tau-mediated neurodegeneration and decreases synaptic phagocytosis by microglia. Neuron (2021)
4. Litvinchuk, A., Huynh, T., Shi, Y., Jackson, R., Finn, M.B., Manis, M., Francis, C.M., Tran, A., Sullivan, P/M., Ulrich, J., Hyman, B.T., Cole, T., Holtzman, D.M. ApoE4 reduction with ASOs decreases neurodegeneration in a tauopathy model. Annals of Neurology (2021)
5. Wong, M., Lewis, M., Doherty, J. J., Shi, Y, Sullivan, P. M., Covey, D. F., Petsko1, G. A., Holtzman, D. M., Paul, S. M., Luo, W. 25-Hydroxycholesterol amplifies microglial IL-1β production in an apoE isoform-dependent manner. Journal of Neuroinflammation. 17(1):192 (2020)
6. Shi, Y., Manis, M., Long, J., Wang, K., Remolina, J., Hoyle, R., Holtzman, D. M. Microglia drive APOE-dependent neurodegeneration in a tauopathy mouse model. Journal of Experimental Medicine 216 (11): 2546-2561. (2019).
Journal of Experimental Medicine Insights: Kemal, S., Vassar, R. Death by microglia. (2019)
7. Shi, Y & Holtzman, D. M. Interplay between innate immunity and Alzheimer’s disease: APOE and TREM2 in the spotlight. Nature Reviews Immunology 18:759-772, (2018)
8. Shi, Y., Yamada, K., Liddelow, S. A., Smith, S. T., Zhao, L., Luo, W., Tsai, R. M., Spina, S., Grinberg, L. T., Rojas, J. C., Gallardo, G., Wang, K., Roh, J., Robinson, G., Finn, M. B., Jiang H., Sullivan, P. M., Baufeld, C., Wood, M. W., Sutphen, C., McCue, L., Xiong, C., Del-Aguila, J. L., Morris, J. C., Cruchaga, C., Fagan, A. M., Miller, B. L., Boxer, A. L., Seeley, W. W., Butovsky, O., Barres, B. A., Paul, S. M., Holtzman, D. M. ApoE4 markedly exacerbates tau-mediated neurodegeneration in a mouse model of tauopathy. Nature 549, 523-527, (2017).
Science News: A new study is changing how scientists think about Alzheimer's disease (2017). https://www.science.org/content/article/new-study-changing-how-scientists-think-about-alzheimer-s-disease
9. Wang, Y., Ulland, T., Ulrich, J., Song, E., Tzaferis, J., Hole, J., Yuan, P., Mahan, T., Shi, Y., Gilfillan, S., Cella, M., Grutzendler, J., DeMattos, R., Cirrito, J., Holtzman, D., Colonna, M. TREM2-mediated early microglial response limits diffusion and toxicity of amyloid plaques. Journal of Experimental Medicine 213, 667-675, (2016).
10. Jia, S., Liu, Y., Shi, Y., Ma, Y., Hu, Y., Wang M., Li, X. Elevation of Brain Magnesium Potentiates Neural Stem Cell Proliferation in the Hippocampus of Young and Aged Mice. Journal of Cellular Physiology 231, 1903-1912, (2016).
11. Achariyar, T., Li, B., Peng, W., Verghese, P., Shi, Y., McConnell, E., Benraiss, A., Kasper, T., Song, W., Takano, T., Holtzman, D., Nedergaard, M., Deane, R. Glymphatic distribution of CSF-derived apoE into brain is isoform specific and suppressed during sleep deprivation. Molecular Neurodegeneration 11, 74 (2016)
12. Frey, E., Valakh, V., Karney-Grobe, S., Shi, Y., Milbrandt, J., DiAntonio, A. An in vitro assay to study induction of the regenerative state in sensory neurons. Experimental Neurology 263, 350-363, (2015)



