
Lin Mei
1977-1982 B.S. in Clinical Medicine Science, Jiangxi Medical College, P.R. China.
1983-1985 M.D. in Neuropharmacology, Academy of Military Medical Sciences, P.R. China.
1986-1989 Ph. D in Pharmacology and Toxicology, University of Arizona, USA.
1989-1994, Postdoctoral, Johns Hopkins University School of Medicine.
1994-1999, Assistant Professor, University of Virginia.
1999-2004, Assistant Professor and Associate Professor, University of Alabama at Birmingham.
2004-2017, Professor and Chair, Medical College of Georgia, Augusta University, Professor and Chair of Department of Neuroscience and Regenerative Medicine
2017-2022, Professor and Chair, Department of Neurosciences, Allen C. Holmes Professor, and Inaugural Director, Cleveland Brain Health Initiative (CBHI), Case Western Reserve University School of Medicine, Cleveland.
2023-present, Professor and Chair of Chinese Institutes for Medical Research (CIMR), Beijing, Distinguished researcher of Chinese Institute for Brain Research (CIBR), Beijing.
The brain contains billions of nerve cells, or neurons, which receive and integrate signals from the environment and govern the body’s responses. Neuronal activity is made possible by synapses, contacts between neurons or between a neuron and a target cell. Neurotransmitter molecules are released from the presynaptic membrane and activate receptors on the postsynaptic membrane, thus establishing neuronal communication. As such, synapses are fundamental units of neural circuitry and enable complex behaviors.
Research in our lab has focused on mechanisms of synapse formation, neurotransmission, and synaptic plasticity. Our goal is to contribute to a better understanding of these processes and the development of potential therapeutic strategies for psychiatric disorders such as schizophrenia, autism, and depression and neurological disorders such as neuromuscular disorders and epilepsy. Over twenty former doctoral students or postdoctoral researchers who have worked in our laboratory now have their own laboratories.

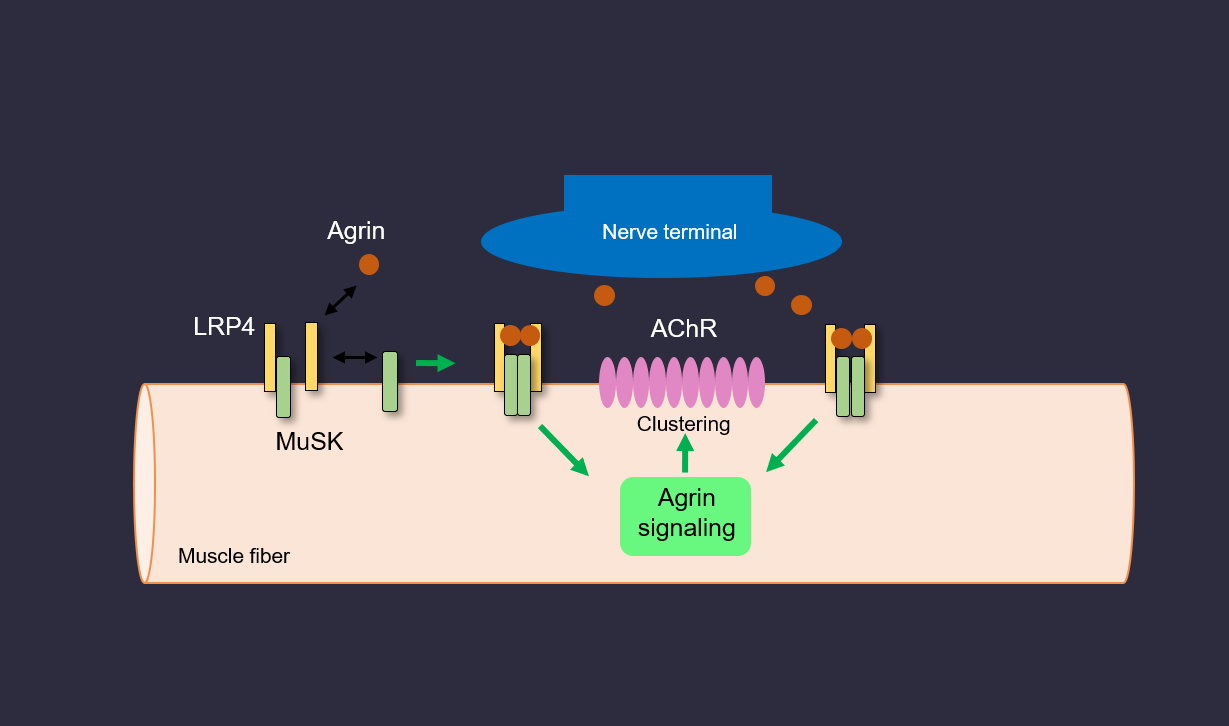
The neuromuscular junction (NMJ) formation and disorders
Our daily activities – getting out of bed, walking, eating, drinking, and sitting – require the proper function of the NMJ, a peripheral synapse between motoneurons and muscle fibers that is critical for accurate and rapid control of muscle contraction. This classic chemical synapse has served as an informative model of synapse structure and function. Although many synaptic components have been identified, how they are assembled to form the NMJ remains unclear. The NMJ formation requires agrin, a trophic factor from motoneurons and MuSK, a transmembrane tyrosine kinase that can be activated by agrin; however, agrin does not interact with MuSK. We identified LRP4 as a receptor for agrin and revealed mechanisms for how signals are transduced from agrin to MuSK by solving a crystal structure of an agrin-LRP4 complex. We have discovered antibodies against LRP4 and agrin in patients with myasthenia gravis and demonstrated that they are causal to myasthenia gravis. These antibodies are used as novel biomarkers for the diagnosis of myasthenia gravis. We demonstrated that rapsyn, a classic scaffold protein, undergoes liquid-liquid phase separation to form membraneless condensates for AChR clustering and NMJ formation. In addition, rapsyn possesses an E3 ligase activity that is required for NMJ formation. We have revealed novel pathways that are critical for the development, maintenance and regeneration of the NMJ.
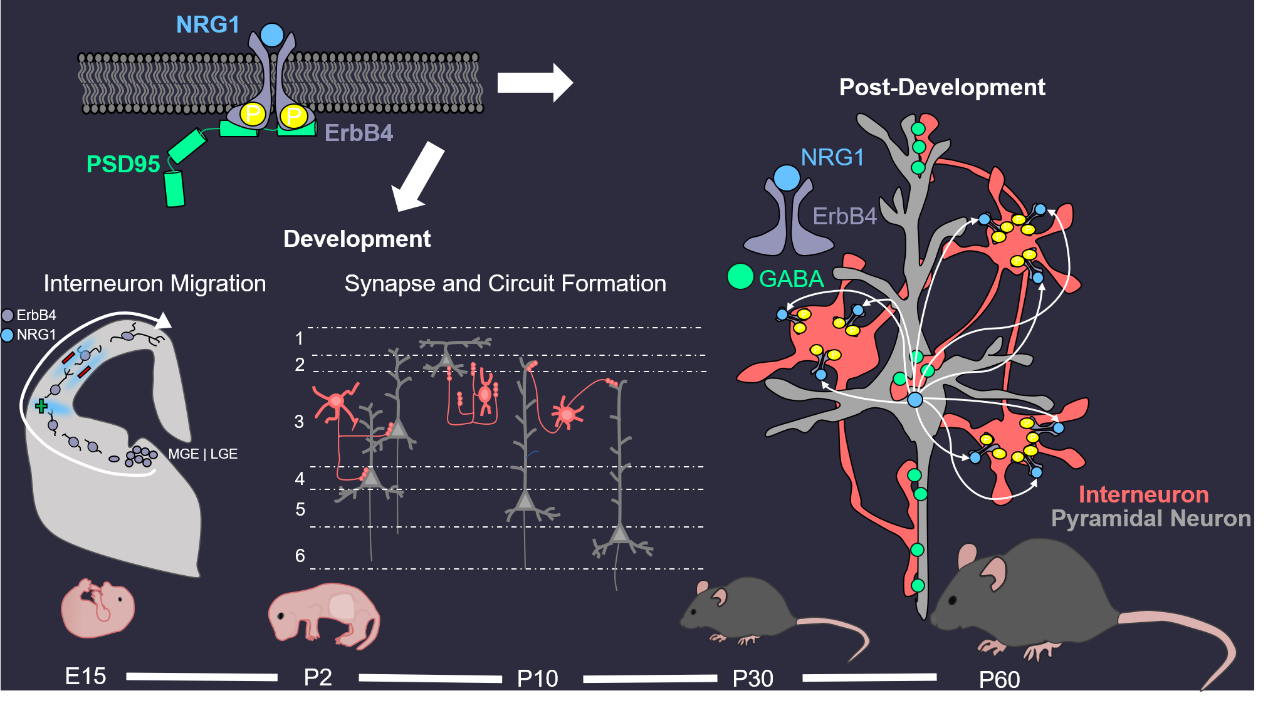
Neuregulin 1 (NRG1) and ErbB4 signaling in excitation-inhibition (E/I) balance
There are two main types of neurons: excitatory (also called projection or pyramidal) neurons that use glutamate as a neurotransmitter and inhibitory interneurons (INs) that release GABA, Comprising 15%–20% of total neurons in the brain, INs control the excitability of projection neurons. Via feedforward and feedback inhibition, interneurons increase the computational power of cortical networks and synchronize both local and distant cortical circuits that are key to oscillatory activity. Disruptions of GABA signaling have been implicated in brain disorders including autism, depression, intellectual disability, Rett syndrome, and schizophrenia. We have demonstrated that NRG1 and its receptor ErbB4 are critical to GABA activity in the brain. NRG1 is produced by pyramidal neurons in an activity-dependent manner. It binds to ErbB4 in INs to promote GABA release and consequently suppresses the activity of pyramidal neurons. This homeostatic pathway is critical to many brain functions including working memory and fear. Translationally, both NRG1 and ErbB4 are risk genes for major depression disorders and schizophrenia. Our research has provided insight into the pathophysiological mechanisms of devastating brain disorders.
Pathological mechanisms of brain disorders
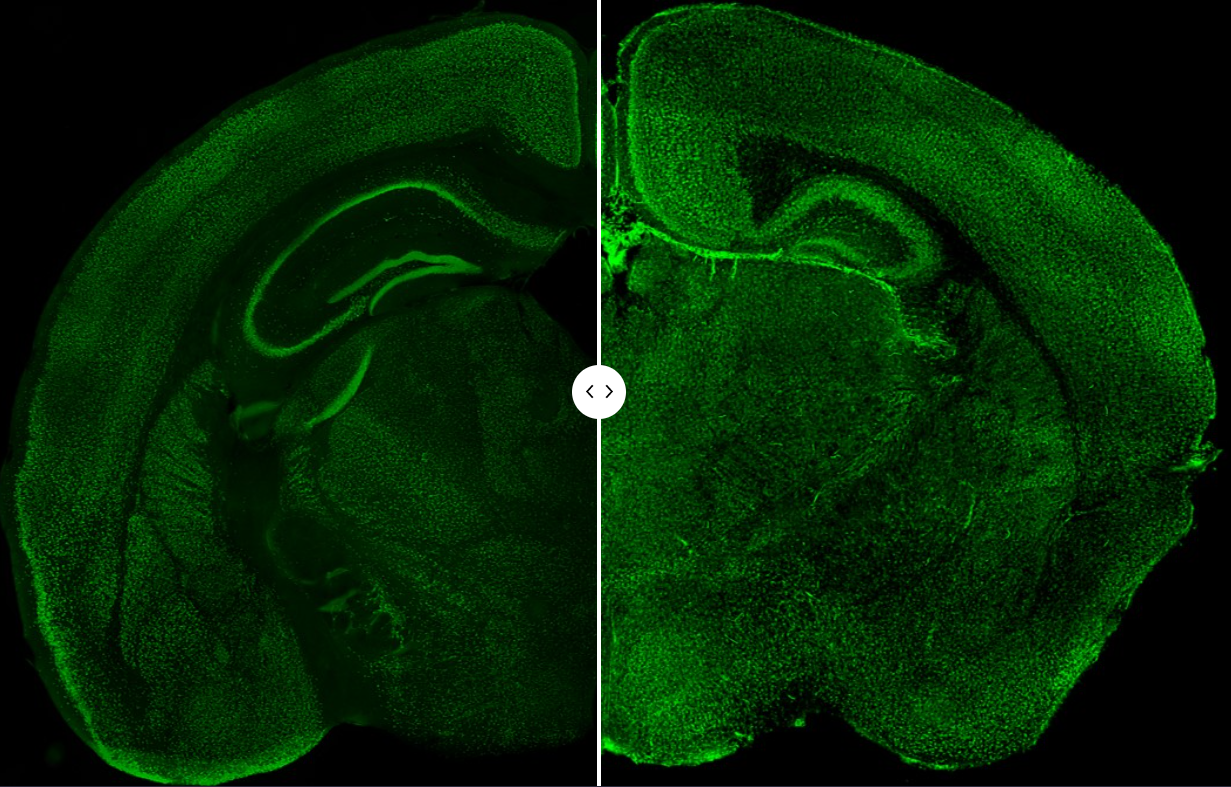
Many psychiatric disorders lack clear pathological hallmarks and thus are poorly understood in terms of pathophysiological mechanisms. With the advance in powerful genetic analysis, risk genes have been identified for various brain disorders. By studying mouse models of deficiency and/or de novo mutations of Erbin, TMEM108, NRG3, Caspr3, and Cullin 3 (images below, left wildtype, right Cullin 3 deficient mice), our studies reveal their physiological functions in neural development and neurotransmission and provide insight into pathophysiological mechanisms of schizophrenia, major depression, and autism.
2023
1. R. Cao*, P. Chen*, H.S. Wang*, H. Jing, H. Zhang, G. Xing, B. Luo, J. Pan, Z. Yu, W.C. Xiong, L. Mei. Intrafusal-fiber LRP4 for Muscle Spindle Formation and Maintenance in Adult and Aged Animals. Nature Communications. 2023. doi.org/10.1038/s41467-023-36454-8. (*co-first).
2. E.D. Kosco*, H. Jing, P. Chen, W.C. Xiong, L. Mei. DOK7 Promotes NMJ Regeneration After Nerve Injury. Molecular Neurobiology. 2023. doi.org/10.1007/s12035-022-03143-4.
2022
1. Z.T. Tan*, Z. Liu*, Y. Liu, H. Robinson, T.W. Lin, W.C. Xiong, L. Mei. An ErbB4-Positive Neuronal Network in the Olfactory Bulb for Olfaction. The Journal of Neuroscience. 43(34): 6518-6535, 2022. doi.org/10.1523/JNEUROSCI.0131-22.2022. PMID: 35853717. (Cover; *co-first).
2. H.S. Wang*, W.B. Chen*, Z.Q. Dong*, G. Xing, W.P. Cui, L.L. Yao, W.J. Zou, H.L. Robinson, Y. Bian, Z. Liu, K. Zhao, B. Luo, N.N. Gao, H.S. Zhang, X. Ren, Z. Yu, J. Meixiong, W.C. Xiong, L. Mei. A novel spinal neuron connection for heat sensation. Neuron. 110(14): 2315-2333.e6, 2022 May 6. doi: 10.1016/j.neuron.2022.04.021. PMID: 35561677. (*co-first)
3. H.L. Robinson, Z. Tan, I. Santiago-Marrero, E.P. Arzola, T.V. Dong, W.C. Xiong and L. Mei. Neuregulin 1 and ErbB4 Kinase Actively Regulate Sharp Wave Ripples in the Hippocampus. The Journal of Neuroscience. 42(3): 390-404, 2022. doi: 10.1523/JNEUROSCI.1022-21.2021. PMID 34844988. (Cover).
2021
1. W.B. Chen*, B. Luo*, N.N. Gao*, H.W. Li, H.S. Wang, L. Li, W.P. Cui, L. Zhang, D. Sun, F. Liu, Z.Q. Dong, H.S. Zhang, H. B. Su, W.C. Xiong, and L. Mei. Neddylation of Nav1.1 is critical for the excitability of interneurons and seizure prevention. J. Clin. Invest. (*co-first) 131(8):e136956. doi.org/10.1172/JCI136956, 2021 (Commentary by S.C. Cannon, Epilepsy channelopathies go neddy: stabilizing NaV1.1 channels by neddylation. JCI 131(8):e148370). PMID: 33651714 / PMCID: PMC8262483.
2. H. Wang*, W. Cui*, W. Chen*, F. Liu, Z. Dong, G. Xing, B. Luo, N. Gao, W.-J. Zou, K. Zhao, H. Zhang, X. Ren, Z. Yu, H.L. Robinson, Z. Liu, W.C. Xiong, L. Mei. The laterodorsal tegmentum-ventral tegmental area circuit controls depression-like behaviors by activating ErbB4 in DA neurons. Molecular Psychiatry. 2021 May 14. doi: 10.1038/s41380-021-01137-7. PMID: 33990773. (Cover, *co-first)
3. G. Xing, H. Jing, Z. Yu, P. Chen, H. Wang, W.C Xiong, and L. Mei. Membraneless condensates by Rapsn phase separation as a platform for neuromuscular junction formation. Neuron. 109:1963-1978, 2021 (Cover and Preview by Bai and Zhang, Neuron, 1907-1909, 2021). PMID: 34033754 / PMCID: PMC8217331.
4. W. Cui*, N. Gao*, Z. Dong, C. Shen, H. Zhang, B. Luo, P. Chen, D. Comoletti, H. Jing, H. Wang, H. Robinson, W.C. Xiong, and L. Mei. In-trans neuregulin3-Caspr3 interaction controls DA axonal bassoon cluster development. Current Biology 2021 Aug 9;31(15):3330-3342.e7. doi: 10.1016/j.cub.2021.05.045. PMID: 34143959.
5. Z. Yu, M. Zhang, H. Jing, P. Chen, R. Cao, P. Pan, B. Luo, Y. Yu, B.M. Quarles, W.C. Xiong, M.H. Rivner, and L. Mei. Characterization of LRP4/agrin Antibodies from a Patient with Myasthenia Gravis. Neurology 2021 Sep 7;97(10):e975-e987. (Preview by Lisak, Neurology Sep 07, 2021; 97 (10)) Doi: 10.1212/WNL.0000000000012463. PMID: 34233932.
2020
1. Z. Dong*, W. Chen*, C. Chen, H. Wang, W. Cui, Z. Tan, H. Robinson, N. Gao, B. Luo, L. Zhang, K. Zhao, W.C. Xiong and L. Mei. CUL3 Deficiency Causes Social Deficits and Anxiety-like Behaviors by Impairing Excitation-Inhibition Balance through the Promotion of Cap-Dependent Translation. Neuron105: 475-490, 2020 (Cover; Preview by Chen & Maher, Neuron, 105:398-399, 2020). PMID:31780330 / PMCID:PMC7007399. Doi: 10.1016/j.neuron.2019.10.035. (*co-first)
2. H. Zhang, W. Chen, Z. Tan, L. Zhang, Z. Dong, W. Cui, K. Zhao, H. Wang, H. Jing, R. Cao, C. Kim, J.G. Safar, W.-C. Xiong and L. Mei. A role of low-density lipoprotein receptor-related protein 4 (LRP4) in astrocytic Aβ clearance. J. Neurosci., 2020 Jul 8;40(28):5347:5361. Doi: 10.1523/jneurosci.0250-20.2020. Epub 2020 May 26. PMID:32457076 / PMCID:PMC7343327 (JC by J. Cavieres-Lepe and N. Stuardo, J. Neurosci. 41:3749-3751).
3. N. Gao, K. Zhao, Y. Cao, X. Ren, H. Jing, G. Xing, W.-C. Xiong, and Lin Mei. A role of lamin A/C in preventing neuromuscular junction decline in mice. J. Neurosci.40:7203-7215, 2020 (Cover). PMID: 32817327 / PMCID: PMC7534915. Doi: 10.1523/JNEUROSCI.0443-20.2020.
4. M.H. Rivner, B.M. Quarles, J.-X. Pan, Z. Yu, J.F. Howard Jr, A. Corse, M.M. Dimachkie, C. Jackson, T. Vu, G. Small, R.P. Lisak, J. Belsh, I. Lee, R.J. Nowak, V. Baute, S. Scelsa, J.A. Fernandes, Z. Simmons, A. Swenson, R. Barohn, R.B. Sanka, C. Gooch, E. Ubogu, J. Caress, M. Pasnoor, H. Xu, L. Mei. Clinical features of LRP4/agrin-antibody – positive myasthenia gravis: A multicenter study. Muscle & Nerve 62:333–343, 2020. PMID: 32483837 / PMCID: PMC7496236. Doi: 10.1002/mus.26985.
5. L. Zhang, H. Jing, H. Li, W. Chen, B. Luo, H-S. Zhang, Z-Q. Dong, L. Li, H-B. Su, W-C. Xiong and L. Mei. Neddylation is critical to cortical development by regulating Wnt/b-catenin signaling. Proc. Nat. Acad. Sci. USA. 117:26448-26459, 2020. PMID: 33020269 / PMCID: PMC7584916 Doi: 10.1073/pnas.2005395117.
2019
1. H. Zhang, A. Sathyamurthy, F. Liu, L. Li, L. Zhang, Z. Dong, W. Cui, X. Sun, K. Zhao, H. Wang, H.-Y. H. Ho, W.C. Xiong, L. Mei. Agrin-Lrp4-Ror2 signaling regulates adult hippocampal neurogenesis in mice. eLife 8:e45303 DOI: 10.7554/eLife.45303, 2019. PMID:31268420 / PMCID:PMC6650252.
2. G. Xing, H. Jing, L. Zhang, Y. Cao, L. Li, K. Zhao, Z. Dong, W. Chen, H. Wang, R. Cao, W.C. Xiong, and L. Mei. A mechanism in agrin signaling revealed by a prevalent Rapsyn mutation in congenital myasthenic syndrome. eLife 8:e49180, 2019. PMID:31549961 / PMCID:PMC6779466 Doi: 10.7554/eLife.49180.
2018
1. Y.-N. Wang, D. Figueiredo, X.-D. Sun, Z.-Q. Dong, W.-B. Chen, W.-P. Cui, F. Liu, H.-S. Wang, H.-W. Li, H. Robinson, E.-K. Fei, B.-X. Pan, B-M Li, W-C Xiong and L. Mei. Controlling of glutamate release by neuregulin3 via inhibiting the assembly of SNARE-complex. Proc. Nat. Acad. Sci. 115:2508-2513, 2018. PMID:29463705 / PMCID:PMC5877931 DOI: 10.1073/pnas.1716322115.
2. T.W. Lin, Z. Tan, A. Barik, D. Yin, E. Brudvik, H. Wang, W.C. Xiong, L. Mei. Regulation of Synapse Development by Vgat Deletion from ErbB4-Positive Interneurons. J. Neurosci. 38:2533-2550, 2018. PMID:29431653 / PMCID:PMC5858595 DOI: 10.1523/JNEUROSCI.0669-17.2018.
3. M. Yan, Z. Liu, E. Fei, W. Chen, X. Lai, B. Luo, P. Chen, H. Jing, J. Pan, M. Rivner, W.C. Xiong, and L. Mei. Induction of anti-agrin antibodies causes myasthenia gravis in mice. Neuroscience, 373:113-121, 2018. PMID:29339325 / PMCID:PMC5942223 Doi: 10.1016/j.neuroscience.2018.01.015.
4. Z. Tan*, H. Robinson*, D. Yin, Y. Liu, F. Liu, H. Wang, T.W. Lin, G. Xing, W.C. Xiong, L. Mei. Dynamic ErbB4 activity in hippocampal-prefrontal synchrony and top-down attention in rodents. Neuron 98:380-393, 2018. PMID:29628188 / PMCID:PMC5909841 Doi: 10.1016/j.neuron.2018.03.018 (*co-first).
5. C. Shen*,**, L. Li**, K. Zhao**, L. Bai, A. Wang, X. Shu, Y. Xiao, J. Zhang, K. Zhang, T. Hui, W. Chen, B. Zhang, W. Hsu, W.-C. Xiong, L. Mei*. Motoneuron Wnts regulate neuromuscular junction development. eLife. 7, 2018 pii: e34625. doi: 10.7554/eLife.34625 (*co-corresponding; **co-first). PMID:30113308.
6. K. Zhao*, C. Shen,*,** L. Li, H. Wu, G. Xing, Z. Dong, W. Chen, H. Zhang, J. Pan, H. Wang, W. Cui, X.D. Sun, S. Li, X. Li, X. Huang, W.C. Xiong, and L. Mei**. Sarcoglycan Alpha Mitigates Neuromuscular Junction Decline in Aged Mice by Stabilizing Lrp4. J. Neurosci.38:8860-8873, 2018. (Cover) (*equal contributing; **co-corresponding). PMID:30171091 / PMCID:PMC6181315 Doi: 10.1523/JNEUROSCI.0860-18.2018.
7. H. Wang,* F. Liu,* W. Chen,* X. Sun, W. Cui, Z. Dong, K. Zhao, H. Zhang, H. Li, G. Xing, E. Fei, B.-X. Pan, B.-M. Li, W.-C. Xiong, Lin Mei. Genetic recovery of ErbB4 in adulthood partially restores brain functions in null mice. PNAS115:13105-13110, 2018. PMID:30498032 / PMCID:PMC6304932 Doi: 10.1073/pnas.1811287115. (*co-first)
Selected Recent Reviews
1. G. Xing, W.C. Xiong, L. Mei. Rapsyn as a signaling and scaffolding molecule in NMJ formation and maintenance. Neurosci. Lett. 731:1350132, 2020
2. M Yan, GL Xing, WC Xiong, L Mei. Agrin and LRP4 antibodies as new biomarkers of myasthenia gravis. Ann. New York Acad. Sci. 1413: 126-135, 2018.
3. L. Li, W.C. Xiong, L. Mei, Neuromuscular junction formation, aging, and disorders. Ann. Rev. Physiol. 2018
4. Mei and K.-A. Nave, A neuregulin and ErbB network in the nervous system. Neuron 83:27-49, 2014
5. Wu, Wen C. Xiong, L. Mei. To build a synapse: Signaling pathways in neuromuscular junction assembly. Development 137:1017-1033, 2010
6. Mei and W.C. Xiong, Neuregulin 1 in neural development, synaptic plasticity and schizophrenia. Nature Rev. Neurosci. 9:437-452, 2008.



