
Woo-ping Ge
2000 B.S. Biochemistry, East China Normal University, China
2005 Ph.D. Neurobiology Institute of Neuroscience, Chinese Academy of Sciences, China
2005-2006 Research Associate, Lab of Shumin Duan, Institute of Neuroscience, Chinese Academy of Sciences
2006 Research Associate, Lab of Zuoren Wang, Institute of Neuroscience, Chinese Academy of Sciences
2006 Visiting Scholar, Lab of Chi-Keung Chan, Institute of Physics Academia Sinica, Taiwan
2006-2011 Post-doc, Developmental Neurobiology, University of California, San Francisco/HHMI, USA
2011-2013 Associate specialist, Lab of Lily Jan, University of California, San Francisco/HHMI
2013-2019 Assistant Professor (tenure-track), Children's Research Institute, Department of Pediatrics
Department of Neuroscience, Department of Neurology & Neurotherapeutics
University of Texas Southwestern Medical Center
12/2019-2025/11 Associate Investigator, Chinese Institute for Brain Research, Beijing
12/2025-present Investigator, Chinese Institute for Brain Research, Beijing
Neurological disorders, such as stroke and brain tumors, affect up to one billion people worldwide. Finding new treatments and understanding how these neurological disorders develop requires a better understanding of the complex interactions that occur in the brain. Our lab’s primary interest is studying interactions between brain vasculature (blood vessels) and the nervous system (glial cells and neurons). By combining electrophysiology and in vivo imaging with genetic methods, we hope to determine how the brain builds the gliovascular and neurovascular network during development and how this network can be damaged as the result of a stroke and then repaired.
Current treatment methods for patients with gliomas are hampered by a poor understanding of the underlying biology. Glial cells are critical for brain metabolism, neuronal protection, and cell-cell communication. As a group with long-term experience in studying the function and development of astrocytes and NG2 glia, we are interested in how gliomas interact with adjacent normal glial cells and how glial cells create a microenvironment that influences glioma cell survival, proliferation, and invasion.
Neuron/Glia-Pericyte Interactions
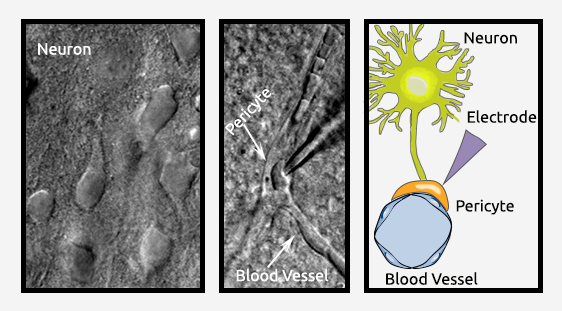
Although pericytes are located along vessels in both the central nervous system and other organs, astrocytic endfeet cover only the vasculature in the central nervous system. It remains unclear whether there are subtypes of pericytes in blood vessels and, if so, what their functions are in the brain. There is also little information available about how different subtypes of pericytes interact with glial cells or neurons in the brain. To answer these questions, we use techniques including electrophysiology and in vivo imaging into the study of brain pericytes. The goal is to isolate pericytes from several sources (arterioles, precapillaries, capillaries, postcapillaries, and venules) to characterize the molecular and cellular profile of pericytes from these different locations. We have already established an electrophysiological technique to record individual pericytes within different segments of blood vessels in acutely isolated brain slices.
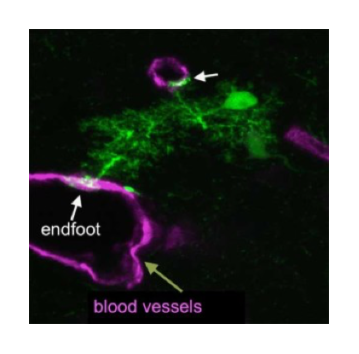
Formation of Gliovascular Interface
Glial cells constitute approximately half of the cells in the human brain. As the largest population of glial cells, astrocytes are crucial for the survival and function of neurons. Together with brain vasculature, astrocytic endfeet form an intricate structure called the gliovascular interface. This interface is critical for the transport of glucose from the blood to neurons, the regulation of cerebral blood flow, and maintenance of the blood-brain barrier. Detachment of astrocytic endfeet from the vascular membrane is responsible for brain edema and results in neurodegeneration. Restoring this function after stroke is critical to improving functional brain recovery in patients. However, it remains unclear how the gliovascular interface forms and develops. We are studying the cellular and molecular mechanisms for interactions between brain vasculature and astrocytes through genetic manipulation and time-lapse slice or in vivo imaging.
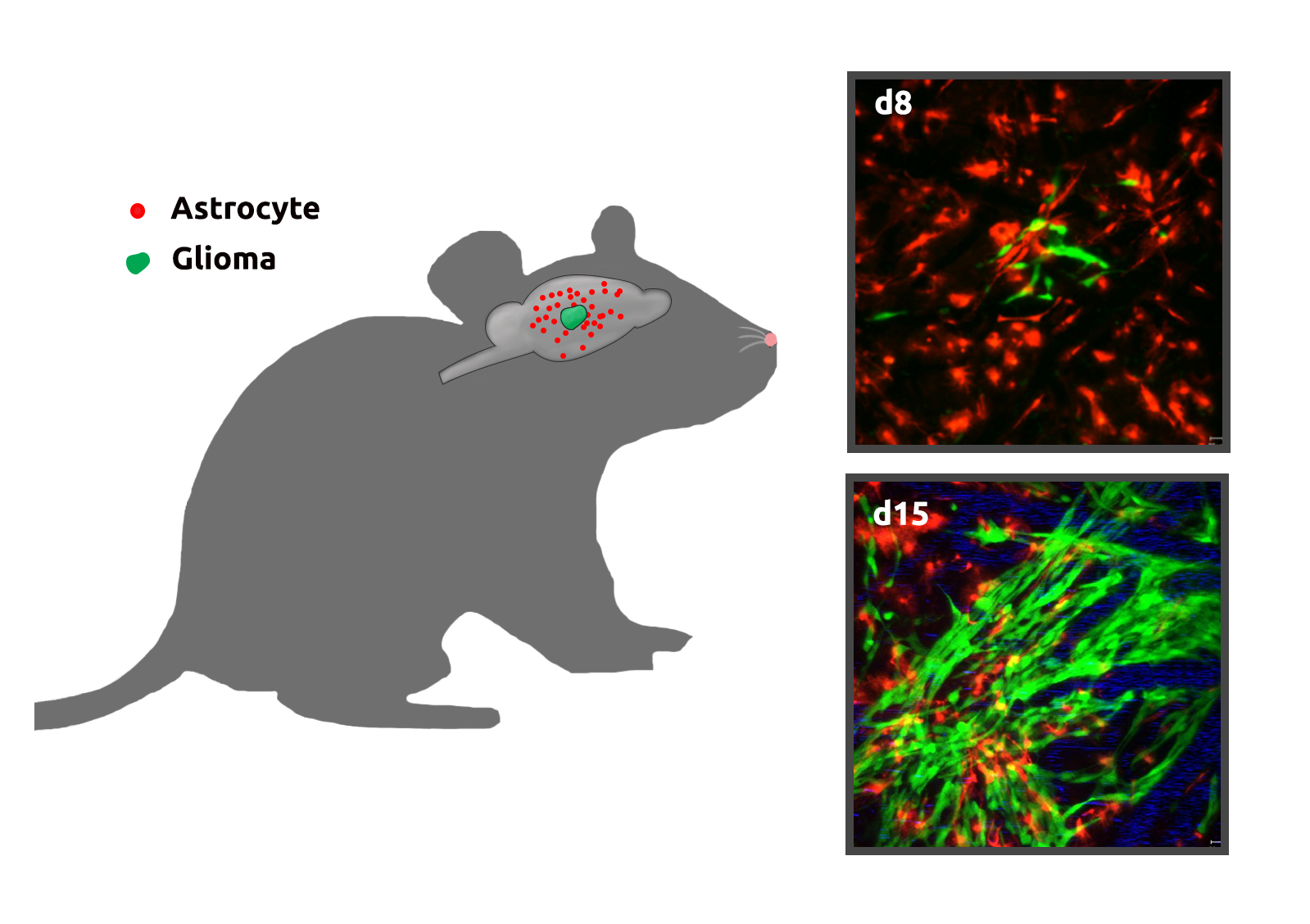
Glia-Glioma Interactions
The brain consists of multiple cell types that form a complex neuron-glia-blood vasculature network. During glioma (brain tumor) development, tumor cells infiltrate normal brain tissue and interact with adjacent stromal cells in this network. The network provides glioma cells with an appropriate environment for colonization, growth, and infiltration. However, the role of normal glial cells, which constitute 50 percent of cells in the human brain and are critical for a number of functions (brain metabolism, neuronal protection, and cell-cell communication), in glioma progression is poorly understood. Improving our understanding of glia-glioma interactions and discovering their underlying mechanisms are critical steps for the diagnosis, prognosis, and treatment of pediatric glioma and are necessary to identify new therapeutic targets.
We are currently investigating whether and how different types of glial cells, especially astrocytes, create a microenvironment that promotes glioma cell survival, proliferation, and invasion. This includes characterization of the fate and potential functional alterations of astrocytes adjacent to gliomas in vivo. We perform longitudinal time-lapse imaging to characterize astrocyte properties (survival, proliferation, and progeny) within and close to gliomas at different developmental stages in order to establish a functional paradigm for glioma growth from its initiation through maturity.
Development of New Tools
We are interested in establishing new tools and methods to study how gliovascular units behave during strokes and in brain tumors in vivo. Currently, vascular surgical challenges present an obstacle for researchers to take advantage of advanced live-imaging technologies in stroke studies, particularly in the developing mouse brain. We have developed a novel approach to induce focal ischemia with precise control of infarct size and occlusion duration in mice at any postnatal age (Jie et al., Nature Methods, 2016). We achieved the occlusion, which is reversible, via micromagnet-mediated aggregation of magnetic nanoparticles within a blood vessel. In combination with longitudinal live imaging, we will investigate the mechanisms underlying disruption and repair of neurovascular units in vivo under ischemic stroke.
2007 Human Frontier Science Program Long-term Fellowship Award
2007 China's Top 10 Advances in Basic Research in 2006
2008 100 Excellent Ph.D. theses of China
2010 The State Natural Science Award (2nd Contributor)
2011 NINDS Pathway to Independence Award (K99/R00)
2017 Bugher-AHA Dan Adams Thinking Outside the Box Award
(The Henrietta B. and Frederick H. Bugher Foundation)
1. Ai D, Qiu B, Chen XJ, Li F, Yao D, Mi H, Li JL, Zhou B, Zuo J, Wang Y, Ge WP, Sun W. Inhibition of microglial Slc2a5 attenuates ischemic brain injury. Metabolism. 2025 Nov 6;175:156429. Epub ahead of print. PMID: 41205649.
2. Yue C, Wang N, Zhai H, Yuan Z, Cui Y, Quan J, Zhou Y, Fan X, Wang H, Wu Z, Mi H, Ge WP, Li Y, Wang X, Luo M. Adenosine signalling drives antidepressant actions of ketamine and ECT. Nature. 2025 Nov 5. Epub ahead of print. PMID: 41193806.
3. Li JL, Bi Z, Chen XJ, Ming T, Qiu B, Li F, Feng Z, Ai D, Zhang T, Wang J, Lin S, Lu Y, Wang Z, Huang J, Zhao F, Zhao H, Wang Y, Sun W, Ge WP. rAAV-miniBEND: A targeted vector for brain endothelial cell gene delivery and cerebrovascular malformation modeling. Nature Biomedical Engineering. 2025 Oct 29. Epub ahead of print. PMID: 41162685.
4. He W, Gao S, Du L, Han T, Yao D, Wu H, Li Q, Li F, Ge WP, Wang Y. Cisternostomy Facilitates Clearance of Metabolic Waste from Cerebrospinal Fluid in Patients with Severe Brain Injury. Aging Dis. 2025 Oct 27.
5. Zhang Z, Pilch J, Lundt S, Zhang N, Chang Y, Singer T, Śladowski D, Hu XL, Zheng L, Ge WP, Zhang H, Li DP, Han X, Ploski R, Ding S. A sensory and motor neuropathy caused by a genetic variant of NAMPT. Sci Adv. 2025 Sep 26;11(39):eadx2407.
6. Gao X, Chen X, Li JL, Ci B, Chen F, Lu N, Shen B, Zheng L, Jia J, Yi Y, Zhang S, Shi Y, Shi K, Propson NE, Huang Y, Poinsatte K, Zhang Z, Yue Y, Bosco DB, Lu YM, Yang SB, Adams RF, Lindner V, Huang F, Wu LJ, Zheng H, Han F, Hippenmeyer S, Stowe AM, Peng B, Margeta M, Wang X, Liu Q, Korbelin J, Trepel M, Lu H, Zhou BO, Zhao H, Sun W, Bachoo RM, Ge WP. Reduction of neuronal activity mediated by blood-vessel regression in the brain. Nature Communications. 2025 Jul 1;16(1):5840.
7. Wang Y, Zhou L, Wang N, Qiu B, Yao D, Yu J, Li T, He M, Xie Y, Yu X, Bi Z, Sun X, Ji X, Li Z, Mo D, Ge WP. Comprehensive characterization of metabolic consumption and production by the human brain. Neuron. 2025;113(11):1708-1722.
8. Ai D, Ming T, Li X, Wang S, Bi Z, Zuo J, Cheng Z, Sun W, Xie M, Li F, Wang X, Qi X, Luan G, Ge WP, Guan Y. Transcriptomic Profiling Unveils EDN3+ Meningeal Fibroblasts as Key Players in Sturge-Weber Syndrome Pathogenesis. Adv Sci (Weinh). 2025 Feb 8:e2408888.
9. Cheng X, Zhao M, Chen L, Huang C, Xu Q, Shao J, Wang HT, Zhang Y, Li X, Xu X, Yao XP, Lin KJ, Xue H, Wang H, Chen Q, Zhu YC, Zhou JW, Ge WP, Zhu SJ, Liu JY, Chen WJ, Xiong ZQ. Astrocytes modulate brain phosphate homeostasis via polarized distribution of phosphate uptake transporter PiT2 and exporter XPR1. Neuron. 2024 Sep 25;112(18):3126-3142.
10. Zhang TT, Ai DS, Wei PL, Xu Y, Bi ZY, Ma FF, Li FZ, Chen XJ, Zhang ZH, Zou XX, Guo ZP, Zhao Y, Li JL, Ye M, Feng ZY, Zhang XS, Zheng LJ, Yu J, Li CL, Tu TQ, Zeng HK, Lei JF, Zhang HQ, Hong T, Zhang L, Luo BY, Li Z, Xing C, Jia CX, Li LJ, Sun WZ, Ge WP. The subcommissural organ regulates brain development via secreted peptides. Nat Neurosci. 2024 Jun;27(6):1103-1115.
11. Yi Y, Li Y, Zhang S, Men Y, Wang Y, Jing D, Ding J, Zhu Q, Chen Z, Chen X, Li JL, Wang Y, Wang J, Peng H, Zhang L, Luo W, Feng JQ, He Y, Ge WP, Zhao H. Mapping of individual sensory nerve axons from digits to spinal cord with the transparent embedding solvent system. Cell Research. 2024; 34(2): 124-139.
12. Zhang D, Ruan J, Peng S, Li J, Hu X, Zhang Y, Zhang T, Ge Y, Zhu Z, Xiao X, Zhu Y, Li X, Li T, Zhou L, Gao Q, Zheng G, Zhao B, Li X, Zhu Y, Wu J, Li W, Zhao J, Ge WP, Xu T, Jia JM. Synaptic-like transmission between neural axons and arteriolar smooth muscle cells drives cerebral neurovascular coupling. Nature Neuroscience. 2024; 27(2): 232-248.
13. Ren J, Huang Y, Ren Y, Tu T, Qiu B, Ai D, Bi Z, Bai X, Li F, Li JL, Chen XJ, Feng Z, Guo Z, Lei J, Tian A, Cui Z, Lindner V, Adams RH, Wang Y, Zhao F, Körbelin J, Sun W, Wang Y, Zhang H, Hong T, Ge WP. Somatic variants of MAP3K3 are sufficient to cause cerebral and spinal cord cavernous malformations. Brain. 2023; 146(9): 3634-3647.
14. Hu Y, Cao K, Wang F, Wu W, Mai W, Qiu L, Luo Y, Ge WP, Sun B, Shi L, Zhu J, Zhang J, Wu Z, Xie Y, Duan S, Gao Z. Dual roles of hexokinase 2 in shaping microglial function by gating glycolytic flux and mitochondrial activity. Nature Metabolism. 2022; 4(12): 1756-1774.
15. Gao X, Li JL, Chen X, Ci B, Chen F, Lu N, Shen B, Zheng L, Jia J, Yi Y, Zhang S, Shi Y, Shi K, Propson NE, Huang Y, Poinsatte K, Zhang Z, Yue Y, Bosco DB, Lu YM, Yang SB, Adams RF, Lindner V, Huang F, Wu LJ, Zheng H, Han F, Hippenmeyer S, Stowe AM, Peng B, Margeta M, Wang X, Liu Q, Korbelin J, Trepel M, Lu H, Zhou BO, Zhao H, Sun W, Bachoo RM, Ge WP . Reduction of neuronal activity mediated by blood-vessel regression in the brain. BioRxiv. (9/15/2020)
16. Xiong N, Gao X, Zhao H, Cai F, Zhang FC, Yuan Y, Liu W, He F, Zacharias LG, Lin H, Vu HS, Xing C, Yao DX, Chen F, Luo B, Sun W, DeBerardinis DJ, Hao Xu H, Ge WP. Using arterial-venous analysis to characterize cancer metabolic consumption in patients. Nature Communications. 2020; 11(1): 3169.
17. Gao X, Zhang Z, Mashimo T, Shen B, Nyagilo J, Wang H, Wang Y, Liu Z, Mulgaonkar A, Hu XL, Piccirillo SGM, Eskiocak U, Davé DP, Qin S, Yang Y, Sun X, Fu YX, Zong H, Sun W, Bachoo RM, Ge WP. Gliomas Interact with Non-glioma Brain Cells via Extracellular Vesicles. Cell Reports. 2020;30(8):2489-2500.
18. Huang G, Zhao T, Wang C, Nham K, Xiong YH, Gao X, Wang Y, Hao G, Ge WP, Sun X, Sumer BD, Gao J. PET imaging of occult tumours by temporal integration of tumour-acidosis signals from pH-sensitive 64Cu-labelled polymers. Nature Biomedical Engineering. 2020; 4: 314-324
19. Huang L, Chambliss KL, Gao X, Yuhanna IS, Behling-Kelly E, Bergaya S, Ahmed M, Michaely P, Luby-Phelps K, Darehshouri A, Xu L, Fisher EA, Ge WP, Mineo C, Shaul PW. SR-B1 drives endothelial cell LDL transcytosis via DOCK4 to promote atherosclerosis. Nature. 2019; 569(7757): 565-569.
20. Jing D, Zhang S, Luo W, Gao X, Men Y, Ma C, Liu X, Yi Y, Bugde A, Zhou BO, Zhao Z, Yuan Q, Feng JQ, Gao L, Ge WP, Zhao H. Tissue clearing of both hard and soft tissue organs with the PEGASOS method. Cell Research. 2018; 28: 803-818.
21. Celen C, Chuang JC, Luo X, Nijem N, Walker AK, Chen F, Zhang S, Chung AS, Nguyen LH, Nassour I, Budhipramono A, Sun X, Bok LA, McEntagart M, Gevers EF, Birnbaum SG, Eisch AJ, Powell CM, Ge WP, Santen GW, Chahrour M, Zhu H. Arid1b haploinsufficient mice reveal neuropsychiatric phenotypes and reversible causes of growth impairment. Elife. 2017; 6: e25730.
22. Jia JM, Chowdary PD, Gao X, Ci B, Li W, Mulgaonkar A, Plautz EJ, Hassan G, Kumar A, Stowe AM, Yang SH, Zhou W, Sun X, Cui B, Ge WP. Control of cerebral ischemia with magnetic nanoparticles. Nature Methods. 2016; 14(2):160-166.
23. Shen Y, Ge WP, Li Y, Hirano A, Lee HY, Rohlmann A, Missler M, Tsien RW, Jan LY, Fu YH, Ptáček LJ. Protein mutated in paroxysmal dyskinesia interacts with the active zone protein RIM and suppresses synaptic vesicle exocytosis. PNAS. 2015; 112(10): 2935-2941.
24. Yu D, Gustafson WC, Han C, Lafaye C, Noirclerc-Savoye M, Ge WP, Thayer DA, Huang H, Kornberg TB, Royant A, Jan LY, Jan YN, Weiss WA, Shu X. An improved monomeric infrared fluorescent protein for neuronal and tumour brain imaging. Nature Communications. 2014; 5: 3626.
25. Ge WP, Miyawaki A, Gage FH, Jan YN, Jan LY. Local generation of glia is a major astrocyte source in postnatal cortex. Nature. 2012; 484 (7394); 376-380.
26. Ultanir SK, Hertz NT, Li G, Ge WP, Burlingame AL, Pleasure SJ, Shokat KM, Jan LY, Jan YN. Chemical genetic identification of NDR1/2 kinase substrates AAK1 and Rabin8 uncovers their roles in dendrite arborization and spine development. Neuron. 2012; 73(6): 1127-1142.
27. Lee HY, Ge WP, Huang W, He Y, Wang GX, Rowson-Baldwin A, Smith SJ, Jan YN, Jan LY. Bidirectional regulation of dendritic voltage-gated potassium channels by the fragile X mental retardation protein. Neuron. 2011; 72(4): 630-642.
28. Ge WP, Zhou W, Luo Q, Jan LY, Jan YN Jan. Dividing glial cells maintain differentiated properties including complex morphology and functional synapses. PNAS. 2009; 106(1): 328-333.
29. Chung HJ, Ge WP, Qian X, Wiser O, Jan YN, Jan LY. G protein-activated inwardly rectifying potassium channels mediate depotentiation of long-term potentiation. PNAS. 2009; 106(2): 635-640.
30. Ge WP, Yang XJ, Zhang Z, Wang HK, Shen W, Deng QD, Duan S. Long-term potentiation of neuron-glia synapses mediated by Ca2+-permeable AMPA receptors. Science. 2006; 312(5779): 1533-1537.
31. Jin W, Ge WP, Xu J, Cao M, Peng L, Yung W, Liao D, Duan S, Zhang M, Xia J. Lipid binding regulates synaptic targeting of PICK1, AMPA receptor trafficking, and synaptic plasticity. The Journal of Neuroscience. 2006; 26(9): 2380-2390.
32. Yang Y, Ge WP, Chen Y, Zhang Z, Shen W, Wu C, Poo M, Duan S. Contribution of astrocytes to hippocampal long-term potentiation through release of D-serine. PNAS. 2003; 100(25):15194-15199.
33. Zhang JM, Wang HK, Ye CQ, Ge WP, Chen Y, Jiang ZL, Wu CP, Poo MM, Duan S. ATP released by astrocytes mediates glutamatergic activity-dependent heterosynaptic suppression. Neuron. 2003; 40(5): 971-982.



