
Jiyan Ma
Ph.D. Biochemistry and Molecular Biology, University of Illinois at Chicago, Chicago, USA
Master of Medicine, Shanghai Medical University, Shanghai, China
Bachelor of Medicine, Shanghai Medical University, Shanghai, China
Investigator, Chinese Institute for Brain Research, Beijing, China
Professor, Van Andel Institute, Grand Rapids, Michigan, USA
Associate professor with tenure, Ohio State University, Columbus, Ohio, USA
Assistant professor, Ohio State University, Columbus, Ohio, USA
Postdoc/Research Associate, Lab of Susan Lindquist, University of Chicago/HHMI, Chicago, Illinois, USA
The overall goal of our research is to 1) elucidate the pathogenic mechanism of protein aggregates in neurodegenerative diseases and 2) develop effective measures against these disorders. Our studies aim to address a huge demand by our aging society: the increased incidence of late-age-onset neurodegenerative disorders, such as Alzheimer’s and Parkinson’s disease. Currently, we do not have a cure or any effective treatment to slow the disease progression. Elucidating the pathogenic mechanism and development of effective prophylactic, diagnostic, and therapeutic strategies are urgently needed.
Although the late-age-onset neurodegenerative diseases have different clinical manifestations, they do share a common property; specifically, they all involve the deposition of protein aggregates in the central nervous system. The correlation of these protein aggregates with the disease is well established, but whether and how these protein aggregates cause neurodegenerative disease remains obscure. Our studies intend to answer these important questions by studying two of these proteins: prion protein and alpha-synuclein.
Alpha synuclein (α-syn) is misfolded and accumulated in a group of neurodegenerative diseases called α-synucleinopathies, which include Parkinson’s disease (PD), Dementia with Lewy Bodies (DLB) and Multiple System Atrophy (MSA). The correlation of misfolded α-syn and neurodegeneration is well supported, but the initiation of α-syn misfolding and the neurotoxic mechanism of misfolded α-syn remain unknown. We are taking a multifaceted approach to investigate the role of α-syn in neurodegenerative diseases. Taking advantage of experimental models based on the seeding activity of preformed α-syn amyloid fibrils, we are studying the initial molecular and cellular changes induced by misfolded α-syn. We are also interested in developing α-syn seeding assay to determine whether the seeding activity of α-syn aggregates can be a reliable and convenient biomarker for α-synucleinopathies.
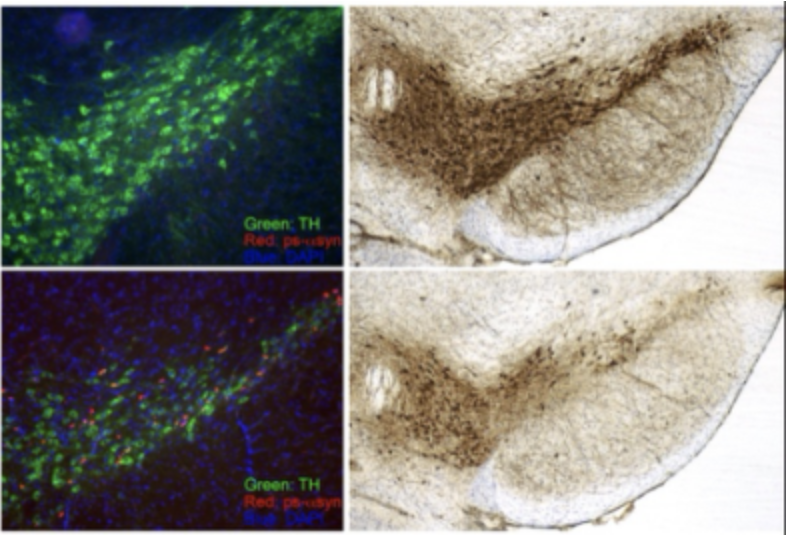
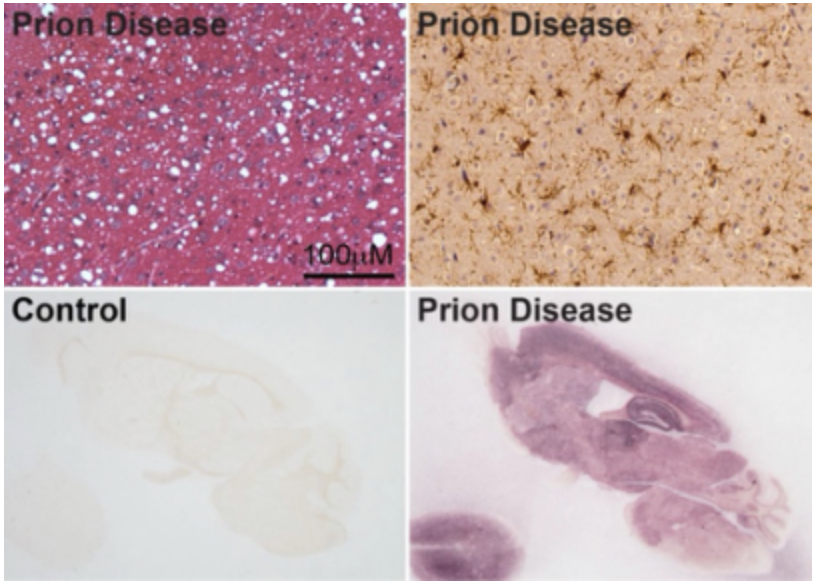
Prion protein (PrP) is a host-encoded glycoprotein that attached to cell membrane via a GPI-anchor. Misfolded PrP is present in a large group of neurodegenerative disease called transmissible spongiform encephalopathies (TSEs) or prion disease, which include Creutzfeldt-Jakob disease in humans, scrapie in sheep, and chronic wasting disease in deer and elk. The uniqueness of this group of neurodegenerative disorders is that they are also bona fide infectious diseases. Our lab has made important contributions to the demonstration that the infectious agent of TSEs is indeed a prion, a misfolded PrP form that aggregates in diseased brain. Currently, we are studying what features of PrP aggregates are essential for the prion’s infectivity and developing strategies to detect and neutralize it. The fact that prion disease is an authentic neurodegenerative disease in rodents is a great advantage in studying the pathogenic changes, and these studies will also shed light on the pathogenic mechanisms of other neurodegenerative disorders. We are also interested in the biological activities of normal PrP in other diseases and determining whether modulating PrP can influence disease progression.
· Graduate School Fellowship Award at University of Illinois at Chicago
· Young Scientist Travel Award, International Symposium on Protein Traffic, Glycosylation and Human Health, Interlaken, Switzerland.
· Ellison Medical Foundation New Scholar in Aging Award
· Excellence in teaching award, College of Medicine, Ohio State University
Board member of Asia Pacific Society of Prion Research (APSPR); Standing and ad hoc member of NIH study sections and program project reviews; member of review panels for UK MRC and BBSRC, Parkinson’s UK, Alberta Prion Research Institute, Alberta Alzheimer Research Program, German-Israeli Foundation for Science Research and Development, Israel Science Foundation, Italian Ministry of Health, Alzheimer’s Association, American Federation for Aging Research.
Selected Publications
Prion
1. Ma Y and Ma J. (2020) Immunotherapy against prion disease. Pathogens. 9(3):216
2. Wang F, Wang X, Orrú C.D., Groveman B. R., Surewicz K., Abskharon R., Imamura M., Yokoyama T., Kim Y.S., Vander Stel K.J., Sinniah K., Priola S.A., Surewicz W.K., Caughey B., Ma J. (2017) Self-propagating, protease-resistant, recombinant prion protein conformers with or without in vivo pathogenicity. PLOS Pathog 13(7): e1006491.
3. Wang X, McGovern G, Zhang Y, Wang F, Zha L, Jeffrey M, Ma J. (2015) Intraperitoneal infection of wild-type mice with synthetically generated mammalian prion PLOS Pathog 11(7):e1004958.
4. Ma J. (2012) The role of Cofactors in Prion Propagation and Infectivity PLOS Pathog 8(4): e1002589.
5. Wang, F.*, Wang, X.*, Yuan, C., Ma, J. (2010) Generating a Prion with Bacterially Expressed Recombinant Prion Protein Science 327:1132-1135. (* equal contribution)
6. Wang, F., Yang, F., Hu, Y., Wang, X., Wang, X., Jin, C., Ma, J. (2007) Lipid interaction converts prion protein to a PrPSc-like proteinase K resistant conformation under physiological conditions Biochemistry. 46(23):7045-7053.
7. Ma, J., Wollmann, R., Lindquist, S. (2002) Neurotoxicity and neurodegeneration when PrP accumulates in the cytosol Science 298(5599):1781-5
8. Ma, J. and Lindquist, S. (2002) Conversion of PrP to a self-perpetuating PrPSc-like conformation in the cytosol Science 298(5599):1785-8
9. Ma, J. and Lindquist, S. (2001) Wild-type and mutant PrP accumulate in the cytoplasm upon proteasome inhibition Proc. Natl. Acad. Sci. USA. 98:14955-14960.
10. Ma. J. and Lindquist, S. (1999) De novo generation of a PrPSc-like conformation in living cells Nat. Cell Biol. 1:358-361.
α-synuclein
1. Bargar C, Wang W, Gunzler SA, LeFevre A, Wang Z, Lerner AJ, Singh N, Tatsuoka C, Appleby B, Zhu X, Xu R, Haroutunian V, Zou W*, Ma J*, Chen SG*. (2021) Streamlined alpha-synuclein RT-QuIC assay for various biospecimens in Parkinson’s disease and dementia with Lewy Bodies. Acta Neuropathol Commun. 9(1):62. (*co-corresponding authors)
2. Wang Z, Becker K, Donadio V, Siedlak S, Yuan J, Rezaee M, Incensi A, Kuzkina A, Orrú CD, Tatsuoka C, Liguori R, Gunzler SA, Caughey B, Jimenez-Capdeville ME, Zhu X, Doppler K, Cui L, Chen SG*, Ma J*, Zou W*. (2020) Skin α-synuclein aggregation seeding activity as a novel biomarker for Parkinson’s disease. JAMA Neurol. 78(1):1-11. (*co-senior authors)
3. Roux A, Wang X, Becker K, Ma J. (2020) Modeling α-synucleinopathy in organotypic brain slice culture with preformed α-synuclein amyloid fibrils. J Parkinsons Dis. 10(4):1397-1410.
4. Marshall LL, Killinger BA, Ensink E, Li P, Li KX, Cui W, Lubben N, Weiland M, Wang X, Gordevicius J, Goetzee GA, Ma J, Jovinge S, Labrie V. (2020) Epigenomic analysis of Parkinson’s disease neurons identifies Tet2 loss as neuroprotective. Nat Neurosci. 23(10):1203-1214.
5. Wang X, Becker K, Levine N, Zhang M, Lieberman AP, Moore DJ, Ma J. (2019) Pathogenic alpha-synuclein aggregates preferentially bind to mitochondria and affect cellular respiration. Acta Neuropathol Commun. 7(1):41.
6. Chen X, Kordich JK, Williams ET, Levine N, Cole-Strauss A, Marshall L, Labrie V, Ma J, Lipton JW, Moore DJ. (2019) Parkinson’s disease-linked D620N VPS35 knockin mice manifest tau neuropathology and dopaminergic neurodegeneration. Proc Natl Acad Sci USA. 116(12):5765-5774.
7. Graham SF, Rey NL, Ugur Z, Yilmaz A, Sherman E, Maddens M, Bahado-Singh RO, Becker K, Schulz E, Meyerdirk LK, Steiner JA, Ma J, Brundin P. (2018) Metabolomic profiling of bile acids in an experimental model of prodromal Parkinson’s disease. Metabolites 8(4):71.
8. Wang B, Underwood R, Kamath A, Britain C, McFerrin MB, McLean PJ, Volpicelli-Daley LA, Whitaker RH, Placzek WJ, Becker K, Ma J, Yacoubian TA. (2018) 14-3-3 proteins reduce cell-to-cell transfer and propagation of pathogenic alpha-synuclein. J Neurosci. 38(38): 8211-8232.
9. Graham SF, Rey NL, Yilmaz A, Kumar P, Madaj Z, Maddens M, Bahado-Singh RO, Becker K, Schulz E, Meyerdirk LK, Steiner JA, Ma J, Brundin P. (2018) Biochemical profiling of the brain and blood metabolome in a mouse model of prodromal Parkinson’s disease reveal distinct metabolic profiles. J Proteome Res. 17(7): 2460-2469.
10. Becker K, Wang X, Vander Stel K, Chu Y, Kordower J, Ma J. (2018) Detecting alpha synuclein seeding activity in formaldehyde-fixed MSA patient tissue by PMCA. Mol Neurobiol. 55(11):8728-8737.



